Product Images Clarithromycin
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 11 images provide visual information about the product associated with Clarithromycin NDC 63304-726 by Ranbaxy Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
125 mg 100's bottle label - clarisusp1

This is a medication called Clarithromycin that comes in the form of an oral suspension. The package contains 100 mL of the medication when reconstituted. It is only available with a prescription. The text also includes identification numbers, such as NDC 63304-821-04. There is some unclear text that cannot be evaluated.*
250 mg 100's bottle label - clarisusp2

This is a label of a medication called Clarithromycin, which is a suspension that is meant to be taken orally. The package contains 100 mL of the medication when reconstituted. The medication is available only with a prescription.*
250 mg 60's bottle label - claritab1

The text is a label for a medication called Clarithromycin, which comes in tablet form and is available only with a prescription. The label also includes a National Drug Code number for the medication.*
500 mg 60's bottle label - claritab2

This is a description of a medication called Clarithromycin. It is in the form of tablets and requires a prescription. The National Drug Code (NDC) for this medication is 63304-726-60. There are 60 tablets per container.*
clinical signs of mac - clinicalsigns
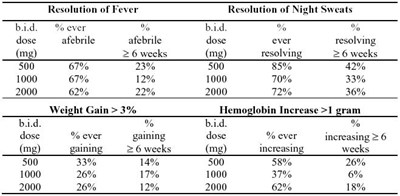
The provided text contains data regarding resolution of fever, night sweats, weight gain, and hemoglobin increase through different doses of medicine administered. For resolution of fever and night sweats at 6 weeks, a 500 mg dose resulted in 23% fever resolution and 85% night sweats resolution. Weight gain of over 3% was observed in 33% of patients taking a 500 mg dose, while hemoglobin increased by over 1 gram in 58% of patients at the same dose and duration. Higher doses of 1000 and 2000 mg also produced positive outcomes, with varying percentages for each outcome.*
eradication rates - eradicatrates
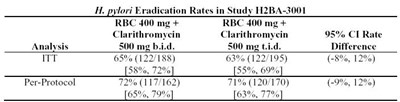
H. pylori Eradication Rates study H2BA-3001 evaluating the efficacy of RBC 400mg + Clarithromycin as compared to RBC 400mg + Clarithromycin, 500 mg b.id. analyses revealed eradication rate ranging from 69% to 72% in ITT (n=188) and 71% to 74% in Per-Protocol (n=117/162) population, respectively. Confidence interval rates were also recorded.*
mic ranges - qcranges
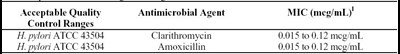
This text provides information on the acceptable quality antimicrobial agents' minimum inhibitory concentration (MIC) range. It includes the MIC range for Clarithromycin and Amoxicillin in meg/mL, tested against H. Pylori ATCC 43504.*
test results - testresults

This appears to be a report on the results of treatment using Clarithromycin for cases involving T pylor po bacteria. The report shows that the treatment was effective in some cases based on the susceptibility test results, but did not eradicate the bacteria in others. The report also shows outcomes for additional treatments involving various medications.*
Susceptibility tests - testwithhpylori
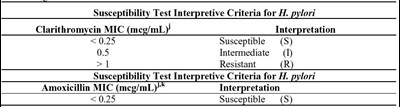
This is a table showing interpretive criteria for susceptibility testing of H. pylori with two antibiotics: Clarithromycin and Amoxicillin. The table indicates the minimum inhibitory concentration (MIC) in mcg/mL for each antibiotic and the corresponding interpretation of susceptibility as either susceptible (S), intermediate (I), or resistant (R).*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

