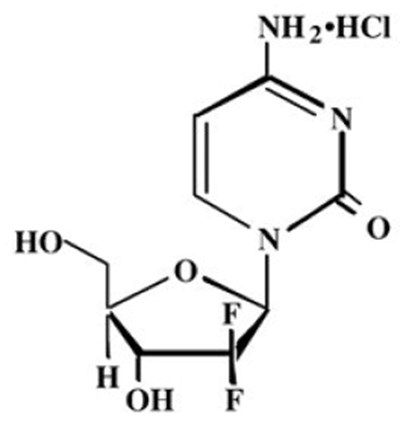
Product Images Gemcitabine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 12 images provide visual information about the product associated with Gemcitabine NDC 63323-126 by Fresenius Kabi Usa, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
gem0j 0002 02
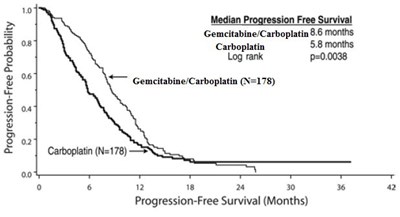
Median Progression Free Survival: Gemcitabine/Carboplatin combination therapy resulted in a median progression-free survival of 8.6 months, while Carboplatin alone had a median progression-free survival of 5.8 months. The Log rank p-value was calculated to be 0.0038. Data for this comparison was obtained from 178 patients for both Gemcitabine/Carboplatin and Carboplatin alone.*
gem0j 0002 03
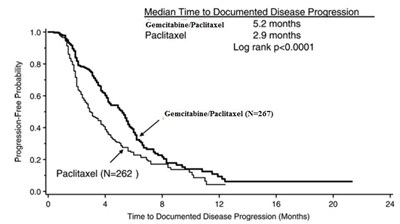
The text provides information on the progression-free probability and median time to documented disease progression for two different treatments - Gemcitabine/Paclitaxel and Paclitaxel. The progression-free probability is shown over time intervals, and the median time to documented disease progression is listed as 5.2 months for Gemcitabine/Paclitaxel and 2.9 months for Paclitaxel. It also includes a Log rank p-value of less than 0.0001, indicating a statistically significant difference between the two treatments in terms of disease progression.*
gem0j 0002 04
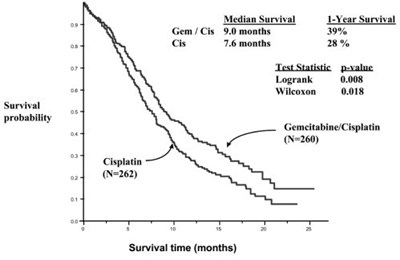
This text provides survival probabilities for two different treatments: Gem/Cis and Cis. It includes information on the survival time in months for each treatment group, as well as the statistical analysis results (Logrank and Wilcoxon) for comparison. The data presented is related to survival time in months for patients receiving Gem/Cis and Cisplatin.*
gem0j 0002 05
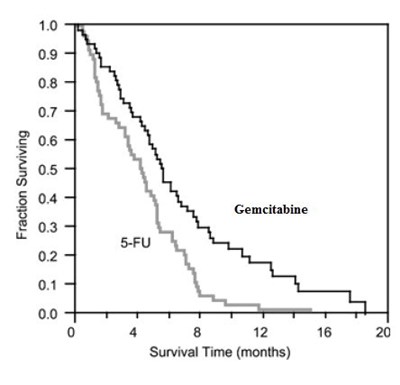
This text provides data on the fraction of patients surviving over time while being treated with Gemcitabine and 5-FU. The graph indicates survival time in months, with several time points (4, 8, 12, 16, and 20 months). It seems to be a chart or graph showing the survival rates of patients undergoing treatment with Gemcitabine and 5-FU over different time intervals.*
gem0j 0002 09

This text appears to be a label for Gemcitabine injection medication. Gemcitabine is used as a cytotoxic agent for intravenous use only. The instructions indicate that to reconstitute the medication, one should mix it with a specific volume of 0.9% Sodium Chloride Injection. The storage conditions and handling precautions are also mentioned in the text. The manufacturer's contact information is provided for product inquiries. It is important to follow the dosage instructions provided in the accompanying package insert.*
gem0j 0002 10

This text contains information about an intravenous medication called Gemcitabine used for injection. It is available in single-dose vials with 1g per vial, and it must be diluted before use. The text also includes a usage warning "For Intravenous use only" and the prescription note "Rx only."*
gem0j 0002 11

This description provides information about Gemcitabine injection, including its composition, dosage instructions, storage recommendations, and cautionary notes. It advises on the reconstitution process, administration, and the importance of discarding unused portions. The text also emphasizes the cytotoxic nature of Gemcitabine and recommends careful handling to avoid exposure. Additionally, it includes details such as the manufacturer's contact information, NDC number, and expiry date. It emphasizes that refrigeration is not recommended and highlights that the product should be used intravenously after proper dilution.*
gem0j 0002 12

This is information about Gemcitabine, a medication used for injection, USP. It is in a 2g per vial dosage for intravenous use only. The text also includes product details like the Single-Dose Vial and prescription information mentioning "Rx only."*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.