Product Images Granisetron
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Granisetron NDC 63323-319 by Fresenius Kabi Usa, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
structure - gran struct
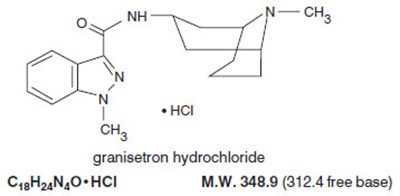
The text contains chemical formulae and compound names for granisetron hydrochloride. It also includes the molecular weight of the compound.*
carton - granisetron 1mg 1 c

Granisetron Hydrochloride Injection, USP is a prescription medication used for intravenous use and comes in a 1 mL single-dose vial. The provided dosage strength is not available.*
vial - granisetron 1mg 1 v

This is a description of Granisetron Hydrochloride Injection, USP with a concentration of 1 mg per mL. It is intended for IV use only and comes in a 1 mL single dose vial. The product does not contain any preservatives.*
carton - granisetron 4mg 1 c

Granisetron Hydrochloride Injection is a medication given through the veins and it is only available with a prescription. The strength of the dose is 4 milligrams per 4 milliliters or 1 milligram per milliliter. It is packaged in a vial that holds 4 milliliters and is designed for multi-dose use. The description ends with an incomplete sentence, possibly due to a scanning error.*
vial - granisetron 4mg 1 v

Granisetron Hydrochloride Injection, USP is a medication used intravenously that contains 4mg per 4 mL (1 mg per mL). It is only for intravenous use and is available in a 4 mL multiple dose vial. This medication is available only with a prescription.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
