Product Images Ceftriaxone
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Ceftriaxone NDC 63323-346 by Fresenius Kabi Usa, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
2gcarton

This is a prescription drug called Ceftriaxone that comes in a box containing 10 single dose vials for intravenous or intramuscular use. The National Drug Code (NDC) for this drug is 63323-347-20 and the lot number is 304720.*
2gvial

The text describes a medicinal product named Ceftriaxone, which is to be used through injection. The product comes in a single-dose vial form and is recommended only for intramuscular or intravenous usage. It is specifically for prescription use. NDC code for this particular Ceftriaxone product is 63323-347-20, and another identifying number is 304720.*
500mgcarton

This is a prescription drug (Rx only) called Ceftriaxone, available in a box containing 25 single-dose vials for intravenous or intramuscular use. The National Drug Code (NDC) for this drug is 63323-345-10 and the lot number is 304510.*
500mgvial

This is a drug label for Ceftriaxone injection, a medication used for intravenous administration. The label provides information about the product's National Drug Code (NDC), dosage strength, and indications for use. It is recommended for use only as prescribed by a medical professional.*
ceftriaxone carton346
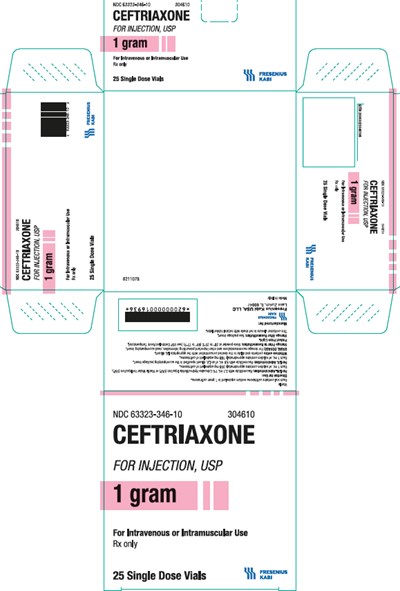
Description: Ceftriaxone is a medication available in the form of 25 single-dose vials with a strength of 1 gram. It is intended for intravenous or intramuscular use and is used in the treatment of certain bacterial infections. The National Drug Code (NDC) for this medication is 63323-346-10.*
ceftriaxone vial346

CEFTRIAXONE is a sterile, prescription-only medication used for injection in LM or LV. Each single dose vial contains 1 gram of ceftriaxone sodium. The powder should be stored between 20°C and 25°C and protected from light. For storage and administration after reconstitution, refer to the package insert. This medication is produced by Fresenius Kabi USA, LLC.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

