Product Images Midazolam
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Midazolam NDC 63323-412 by Fresenius Kabi Usa, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
vial - midazolam vlbl

This product contains 500 mg of Reonly per 100 mL (5mg per ml). It is a pharmacy bulk package meant for non-direct infusion and is sterile. Suitable for intramuscular and intravenous use. Refer to the code for specific information.*
ph fig - ph fig
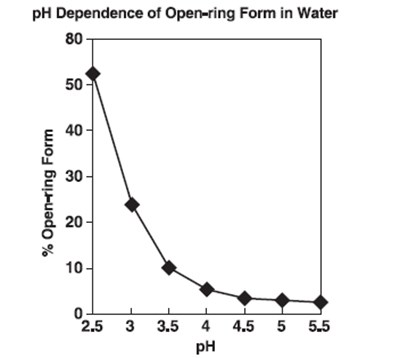
This text seems to refer to a graph or plot showing the pH dependence of the open-ring form in water, with percentages of the open-ring form ranging from 10% to 50%. The graph likely illustrates how the percentage of the open-ring form changes at different pH levels.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




