Product Images Nesacaine Mpf
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Nesacaine Mpf NDC 63323-477 by Fresenius Kabi Usa, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
nes01 0000 04

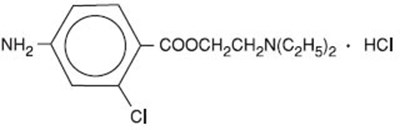
Nesacaine® is a chloroprocaine HCI Injection, USP, with 300mg per 30mL or 10 mg per mL concentration. It is intended for infiltration and nerve block only, and not for use in caudal, epidural, or spinal anesthesia. The product is supplied in a 30mL multiple dose vial and contains Methylparaben. It is available only with a prescription (Rx only).*
nes01 0000 05

NDC 63323-475-37 is a drug called Nesacaine which is used for infiltration and nerve block. It comes in a pack of 25 multiple dose vials containing 30 mL each, and each mL is made up of Chloroprocaine HCI 10mg, Sodium chloride 6.7 mg, Disodium EDTA dihydrate 0.111mg, Methylparaben 1mg. The usual dosage is mentioned in the insert and it is not for caudal, epidural or spinal anghesia. It also states that the drug should not be autoclaved, and should be discarded if the solution is discolored. Furthermore, the drug should be protected from light and stored between 20° to 25°C (68° to 71°F).*
nes01 0000 06

Nesacaine is an injection used for infiltration and nerve block, delivering 600mg of chloroprocaine HCI in a 30mL multiple-dose vial with 20ng of methylparaben per mL. It is labeled for use with infiltration and nerve blocks only and not recommeded for caudal, epidural, or spinal anesthesia. The NDC (National Drug Code) number is 63323-476-01, and the product is intended for prescription use only.*
nes01 0000 07

Nesacaine Injection is a sterile, nonpyrogenic 2% chloroprocaine HCI injection solution designed for infiltration and nerve block use only. It contains 600mg per 30mL and is not intended for caudal, epidural or spinal anesthesias. The solution includes methylparaben and is sold only in an unbroken package. It should not be autoclaved, and the solution should be discarded if discolored. The injection must be protected from light, and the storage conditions should be maintained between 20° to 25°C (68° to 77°F).*
nes01 0000 08

Nesacaine MPF is a chloroprocaine HCI Injection used for infiltration, nerve block, caudal and epidural anesthesia. It is not suitable for spinal anesthesia and comes in a single dose vial of 20 mL with 0 400 mg per 20 mL and 2% concentration. The medication is free of Methylparaben and EDTA. Only available with a prescription.*
nes01 0000 09

Nesacaine-MPF is an injectable solution for Infiltration, Nerve Block, Caudal, and Epidural Anesthesia. This drug is not for Spinal Anesthesia; it is Methylparaben Free and EDTA Free. It is a clear and sterile solution with a pH range from 2.7 to 4.0. Each mL contains Chloroprocaine HCI, Sodium chloride, Sodium hydroxide and/or hydrochloric acid. The correct dosage is indicated in the insert; any unused portion should be discarded. The solution should not be autoclaved, and the seller can only sell it in an unbroken package. It must be protected from light and stored at a temperature range of 20° to 25°C (68° to 77°F).*
nes01 0000 10

Nesacaine MPF is a chloroprocaine HCI injection solution containing 600mg per 20mL for the purpose of infiltration, nerve block, caudal, and epidural anesthesia. It is not recommended for spinal anesthesia, and is methylparaben and EDTA free. It is a single dose vial that requires a prescription. NDC 63323-478-01 and 470827 are also listed.*
nes01 0000 11

This is a product description of Chloroprocaine HCI Injection, USP for Infiltration, Nerve Block, Caudal and Epidural Anesthesia. It comes in single-dose vials of 20 mL, with 30mg Chloroprocaine HCI per mL. It is sterile and non-pyrogenic and should be discarded if the solution is discolored. It needs to be stored at 20°C to 25°C (68°F to 77°F) and protected from light, sodium hydroxide, and hydrochloric acid to adjust pH to 2.7 to 3.5. This product is not intended for spinal anesthesia and should not be autoclaved. Methylparaben free and filled under nitrogen. A usual dosage is inside the insert.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.