Product Images Enoxaparin Sodium
View Photos of Packaging, Labels & Appearance
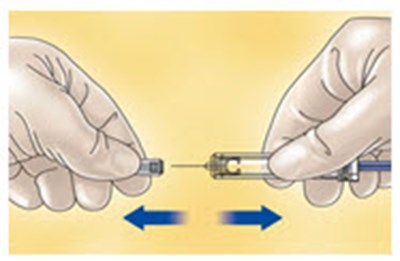
- Figure A - enoxaparin fk 01
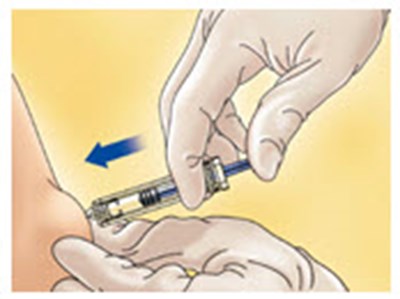
- Figure B - enoxaparin fk 02
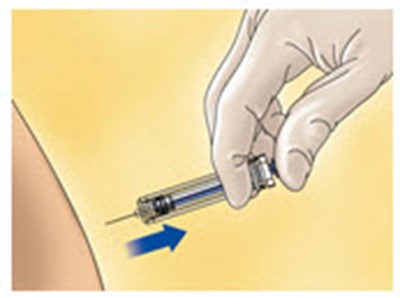
- Figure C - enoxaparin fk 03
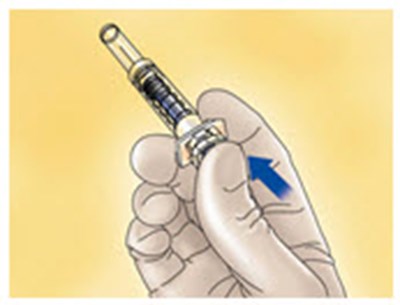
- Figure D - enoxaparin fk 04
- Figure E - enoxaparin fk 05
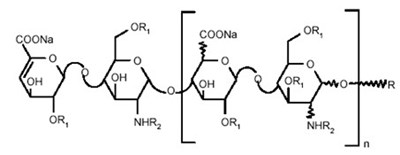
- Chemical Structure - enoxaparin fk 06
- Chemical Structure - enoxaparin fk 07
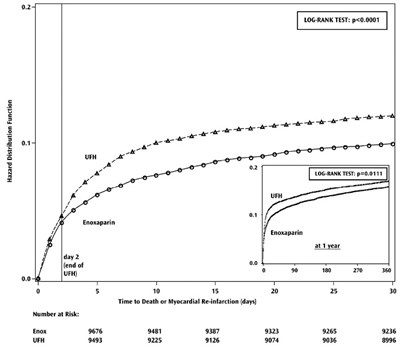
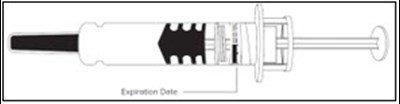
- Figure 1 - enoxaparin fk 08
- Figure 2 - enoxaparin fk 09
- Figure - enoxaparin fk 10
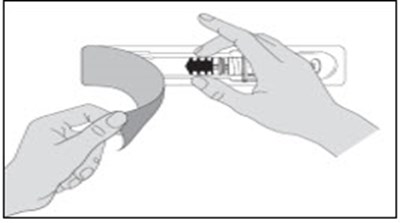
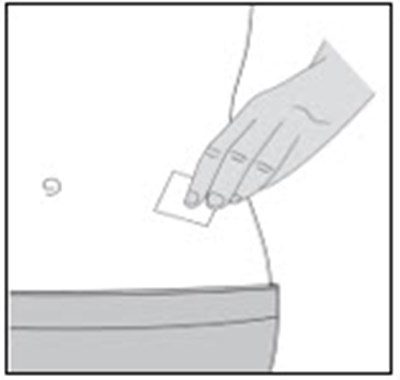
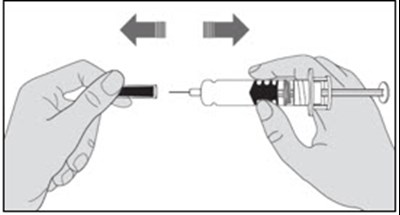
- Figure A - enoxaparin fk 11
- Figure B - enoxaparin fk 12
- Figure C - enoxaparin fk 13
- Figure D - enoxaparin fk 14
- Figure E - enoxaparin fk 15
- Figure F - enoxaparin fk 16
- Figure G - enoxaparin fk 17
- Figure H - enoxaparin fk 18
- Figure I - enoxaparin fk 19
- Figure J - enoxaparin fk 20
- Figure K - enoxaparin fk 21
- Figure L - enoxaparin fk 22
- Figure M - enoxaparin fk 23
- Figure N - enoxaparin fk 24
- PRINCIPAL DISPLAY PANEL - 30 mg/0.3 mL Syringe Carton - enoxaparin fk 25
- PRINCIPAL DISPLAY PANEL - 40 mg/0.4 mL Syringe Carton - enoxaparin fk 26
- PRINCIPAL DISPLAY PANEL - 60 mg/0.6 mL Syringe Carton - enoxaparin fk 27
- PRINCIPAL DISPLAY PANEL - 80 mg/0.8 mL Syringe Carton - enoxaparin fk 28
- PRINCIPAL DISPLAY PANEL - 100 mg/1 mL Syringe Carton - enoxaparin fk 29
- PRINCIPAL DISPLAY PANEL - 300 mg/3 mL Vial Carton - enoxaparin fk 30
- PRINCIPAL DISPLAY PANEL - 120 mg/0.8 mL Syringe Carton - enoxaparin fk 31
- PRINCIPAL DISPLAY PANEL - 150 mg/1 mL Syringe Carton - enoxaparin fk 32
Product Label Images
The following 32 images provide visual information about the product associated with Enoxaparin Sodium NDC 63323-537 by Fresenius Kabi Usa, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - enoxaparin fk 08

This text appears to be a data table with various criteria related to a medical study or clinical trial, including information about subgroups based on sex, age, infarct location, diabetes status, prior MI, fibrinolytic agent used, time to treatment, and clinical outcomes at 30 days. The table also presents relative risk percentages for different groups of patients when comparing Enoxaparin and UFH (heparin) as treatments. The data seems to be evaluating the effectiveness of these treatments based on the specified criteria.*
PRINCIPAL DISPLAY PANEL - 30 mg/0.3 mL Syringe Carton - enoxaparin fk 25

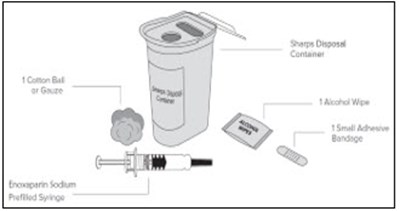
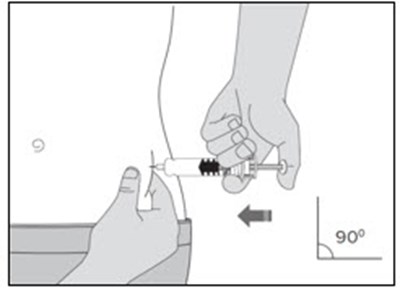
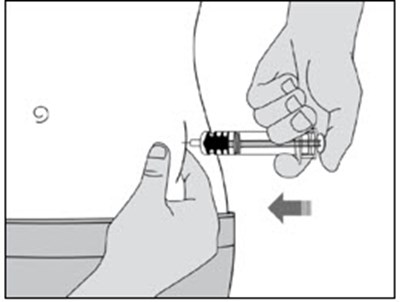
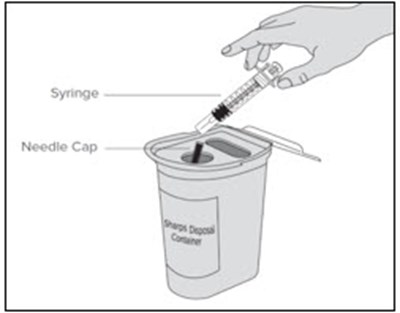
This is a product description for "Enoxaparin Sodium Injection." It is a medication supplied in single-dose syringes with an automatic safety device for subcutaneous injection. Each syringe contains 30 mg of enoxaparin sodium derived from porcine intestinal mucosa in Water for Injection. The dosage and administration instructions, as well as warnings, storage conditions, and directions for use are provided in the text. The safety system of the syringe is detailed, and proper disposal after use is emphasized. The text also mentions the manufacturer, Fresenius Kabi, and various identification numbers associated with the product.*
PRINCIPAL DISPLAY PANEL - 40 mg/0.4 mL Syringe Carton - enoxaparin fk 26

This text is a description of Enoxaparin Sodium Injection in single-dose syringes with automatic safety device for subcutaneous injection. Each syringe contains 40 mg of Enoxaparin Sodium derived from porcine intestinal mucosa. It provides dosage and administration instructions, along with warnings and storage information. The text also includes directions for using the syringe, emphasizing safety precautions and proper disposal. The product is manufactured by Fresenius Kabi USA, LLC.*
PRINCIPAL DISPLAY PANEL - 60 mg/0.6 mL Syringe Carton - enoxaparin fk 27

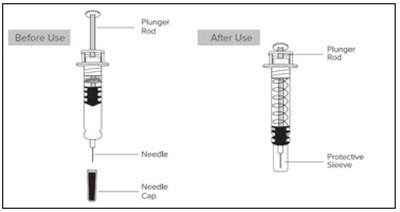
This is a description for Enoxaparin Sodium Injection in single-dose syringes with automatic safety device for subcutaneous injection. Each syringe contains 60 mg of Enoxaparin Sodium derived from porcine intestinal mucosa in Water for Injection. The dosage information is available in the package insert, with each 0.025 mL graduation equaling 2.5 mg. The directions for use include removing the needle shield, adjusting the dose, activating the safety system, injecting following standard technique, and disposing of the syringe in a sharps container. This injection should be stored at 15-30°C (59-86°F). Manufacturer: Fresenius Kabi USA, LLC. The safety system can only be activated once and caution should be taken to avoid accidental needle sticks.*
PRINCIPAL DISPLAY PANEL - 80 mg/0.8 mL Syringe Carton - enoxaparin fk 28

This text provides detailed information about Enoxaparin Sodium Injection, a medication delivered through single-dose syringes with automatic safety devices for subcutaneous injection. It includes instructions on dosage, administration, storage, and safety precautions. The medication is derived from porcine intestinal mucosa and is available in syringes containing 80 mg of Enoxaparin Sodium Injection. The text also outlines the procedure for using the syringe with the safety device and emphasizes proper disposal after use. Manufacturer details and activation warnings for the safety system are also included.*
PRINCIPAL DISPLAY PANEL - 100 mg/1 mL Syringe Carton - enoxaparin fk 29

This text provides information about single-dose syringes with automatic safety devices for subcutaneous injection of Enoxaparin Sodium. It includes details about dosage, administration, storage instructions, and disposal guidelines to ensure safe use. The product is manufactured by Fresenius Kabi, and it is emphasized to keep it out of reach of children. The safety system should be activated only once the syringe has been emptied, and caution is advised to avoid splatter during activation. Proper disposal of the syringe into a sharps container is recommended. Temperature storage guidelines are also mentioned.*
PRINCIPAL DISPLAY PANEL - 300 mg/3 mL Vial Carton - enoxaparin fk 30

This is a description of Enoxaparin Sodium Injection, a medication used for subcutaneous or intravenous injection. Each 0.1mL contains 10 mg of enoxaparin sodium. It is important to follow the dosage information and directions for use provided in the package insert. The medication should be stored at 25°C (77°F) with excursions permitted to 15-30°C (59-86°F). Once opened, the multiple dose vials should not be stored for more than 28 days. This product is manufactured by Fresenius Kabi USA, LLC and is intended for prescription use only.*
PRINCIPAL DISPLAY PANEL - 120 mg/0.8 mL Syringe Carton - enoxaparin fk 31

This is a description of Enoxaparin Sodium Injection packaged in single-dose syringes with an automatic safety device used for subcutaneous injections. Each syringe contains 120 mg of Enoxaparin Sodium derived from porcine intestinal mucosa in Water for Injection. The dosage information is provided in the package insert. The safety system of the syringe is activated by pushing the plunger rod after the injection is complete. It is essential to dispose of the syringe in a sharps container immediately after use. The needle shield must be removed before administering the medication, and the system can only be activated once the syringe is empty. Care should be taken to prevent minimal splatter of fluid while activating the safety system.*
PRINCIPAL DISPLAY PANEL - 150 mg/1 mL Syringe Carton - enoxaparin fk 32

This text describes single-dose syringes with an automatic safety device for subcutaneous injection. The syringes contain Enoxaparin Sodium Injection and come in packages of ten 1mL syringes. The dosage and administration instructions are provided, emphasizing the importance of handling the syringe properly to activate the safety system. It also gives directions on how to use the syringe and advises on storage and disposal after use.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.