Product Images Aptiom
View Photos of Packaging, Labels & Appearance
- PRINCIPAL DISPLAY PANEL – 200 MG SAMPLE WALLET - apt03 0010 12
- PRINCIPAL DISPLAY PANEL – 400 MG SAMPLE WALLET - apt03 0010 13
- PRINCIPAL DISPLAY PANEL – 600 MG SAMPLE WALLET - apt03 0010 14
- PRINCIPAL DISPLAY PANEL – 800 MG SAMPLE WALLET - apt03 0010 15
- PRINCIPAL DISPLAY PANEL – TITRATION SAMPLE PACK WALLET - apt03 0010 16
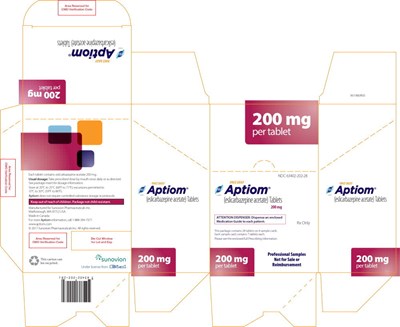
- PRINCIPAL DISPLAY PANEL – 200 MG SAMPLE CARTON - apt03 0010 17
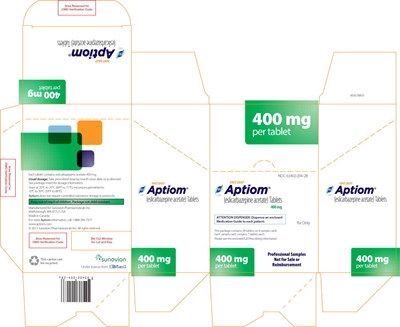
- PRINCIPAL DISPLAY PANEL – 400 MG SAMPLE CARTON - apt03 0010 18
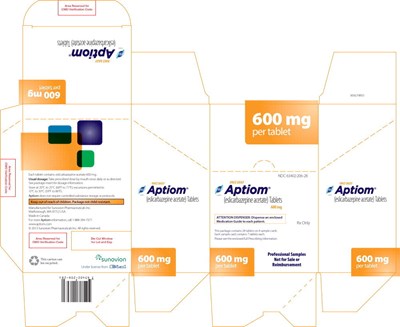
- PRINCIPAL DISPLAY PANEL – 600 MG SAMPLE CARTON - apt03 0010 19
- PRINCIPAL DISPLAY PANEL – 800 MG SAMPLE CARTON - apt03 0010 20
- PRINCIPAL DISPLAY PANEL – TITRATION SAMPLE PACK CARTON - apt03 0010 21
- apt03 0012 08
- apt03 0012 09
- apt03 0012 10
- apt03 0012 11
- apt03 0013 01
- apt03 0013 02
- apt03 0013 03
- apt03 0013 04
- apt03 0013 05
- apt03 0013 06
- apt03 0013 07
Product Label Images
The following 21 images provide visual information about the product associated with Aptiom NDC 63402-202 by Sumitomo Pharma America, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL – 200 MG SAMPLE WALLET - apt03 0010 12

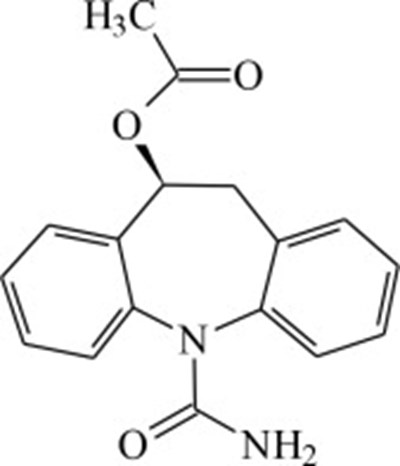
This package contains a medication named Aptiom (eslicarbazepine acetate) in tablet form with a usual dosage of taking it by mouth. The package contains 7 tablets with a dosage of 200mg per tablet. The medication is intended for professional sample use only and is not for sale or reimbursement. The text also includes some unreadable characters and a code for GMO verification.*
PRINCIPAL DISPLAY PANEL – 400 MG SAMPLE WALLET - apt03 0010 13

This is a description of a medication package for Aptiom, which contains tablets of 400mg of eslicarbazepine acetate, manufactured by Sunovion. The package also includes a medication guide which is to be given to each patient.*
PRINCIPAL DISPLAY PANEL – 600 MG SAMPLE WALLET - apt03 0010 14

This is a medication called "Aptiom" which contains tablets of eslicarbazepine acetate. The medication is supposed to be taken orally once a day as per the prescribed dosage or as directed by a medical professional. The tablets should be stored at a temperature range of 20°C to 25°C and should be kept in a non-varnish area. The package contains seven tablets and is not intended for sale or reimbursement purposes. There is additional information on dosage and precautions included in the enclosed Medication Guide.*
PRINCIPAL DISPLAY PANEL – 800 MG SAMPLE WALLET - apt03 0010 15

This is a label of medication named Aptiom (Eslicarbazepine Acetate) Tablets of 800mg. The package contains 7 tablets, and the medication is prescribed only. The label also includes certain instructions for the dispenser.*
PRINCIPAL DISPLAY PANEL – 800 MG SAMPLE CARTON - apt03 0010 20

This appears to be information related to a medication called Aptiom, specifically the 800 mg dosages available in tablet form. It also mentions the active ingredient, eslicarbazepine acetate. However, without additional context or formatting, it is difficult to provide a more comprehensive description.*
PRINCIPAL DISPLAY PANEL – TITRATION SAMPLE PACK CARTON - apt03 0010 21

The text seems to be a mix of gibberish and specific medical terms, drug names and dosages. It appears to be a list of tablets and packs of medication, along with a warning against selling or reimbursement. However, there isn't enough coherent information to provide a useful description.*
apt03 0013 03

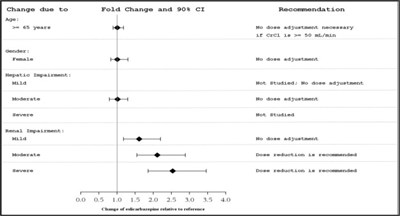
This text appears to be a list of different medications, doses, and recommendations related to changes in dosage and fold change. It also includes a graph with numbers on the x-axis and y-axis, indicating a change in "slcarbazepine" relative to a reference. There is a mention of "Caspemsepinn," "Gabapentin," "Lamotrigine," "Leveticacetn," "Pranepasbitas," "AFTION," "Topsrumate," "vaspronce," "Fhanobarbital," and "phencharbital-like ADs (s.g., primidone)." There is not enough information to understand the context or purpose of this information.*
apt03 0013 04

This appears to be a section of a medical report discussing changes in the medications carbamazepine, gabapentin, Brenyeon, and topiramate. The report mentions "Fold Change and 90% CI" but does not provide any specifics on what these changes are. The report also makes a recommendation to adjust the dose of one of the medications and monitor the concentration of another medication. There is a graph showing changes in other drugs relative to a reference point, potentially relating to pharmacodynamic interaction.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.