Product Images Ibrance
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Ibrance NDC 63539-688 by U.s. Pharmaceuticals, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 3 - ibrance 04
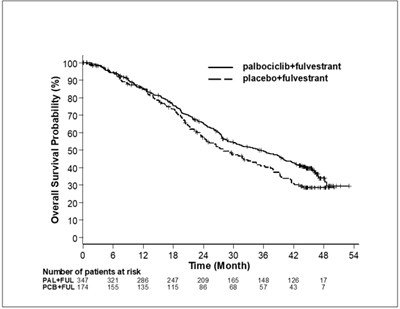
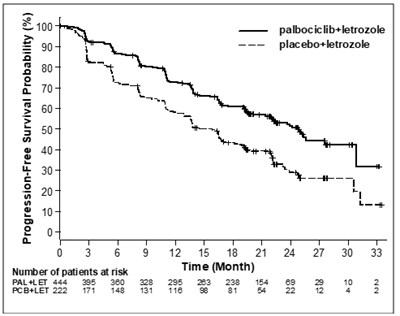
The text describes a graph showing the overall survival probabilities for two treatments. One is a combination of palbociclib and fulvestrant, while the other treatment is a placebo and fulvestrant. The graph has a time axis displaying months and the probability axis displaying values from 0 to 100%. There are also the number of patients at risk displayed. Therefore, the text can be used to understand the benefits of the two treatments for medical purposes.*
Logo - ibrance 05

This is a statement indicating that Pfizer Labs, which is a division of Pfizer Inc, located in New York City, is the distributor of a particular product.*
Logo - ibrance 06

This text is a brief description of the distributor of a pharmaceutical product. It indicates that Pfizer Labs, a division of Pfizer Inc, which is located in NY, NY 10017, is responsible for distributing the product.*
PRINCIPAL DISPLAY PANEL - 75 mg Tablet Dose Pack - ibrance 07

Take IBRANCE for 7 days followed by 7 days off treatment. For more information, visit www.IBRANCE.com. This is a dosage and usage guide for a 75mg blister pack of Palbociclib tablets. The tablets are distributed by US Pharmaceuticals, Pfizer Inc., NY. This is a professional sample and not for sale.*
PRINCIPAL DISPLAY PANEL - 100 mg Tablet Dose Pack - ibrance 09

This text provides dosage and usage information for a medication called IBRANCE, which comes in tablet form containing 100mg of palbociclib. The tablets are stored in their original blister pack and should be kept between 20-25°C (68-77°F). The recommended dose is 1 tablet per day for 21 consecutive days, followed by 7 days off treatment. This information is intended for medical professionals and is not a product for sale to the public. For more information, go to the website www.jibrance.com.*
PRINCIPAL DISPLAY PANEL - 125 mg Tablet Dose Pack - ibrance 11

Take IBRANCE for seven days followed by seven days off treatment. Dosage and usage information can be found in the accompanying prescribing and patient information. Each tablet contains 125mg of palbociclib and should be stored in the original blister pack between 20°C to 25°C (68°F to 77°F), with excursions permitted between 15°C to 30°C (59°F to 86°F). IBRANCE is distributed by US Pharmaceuticals in NY. This is a professional sample and not for sale.*
PRINCIPAL DISPLAY PANEL - 125 mg Tablet Dose Pack Box - ibrance 12

The text is describing a medication named IBRANCE (palbociclib) in the form of tablets of dosage 125mg. It comes in a month box consisting of weekly packs, each pack comprising of 7 IBRANCE tablets. The rest of the text consists of alphanumeric codes that are not clear or readable.*
PRINCIPAL DISPLAY PANEL - Topper Card - ibrance 13
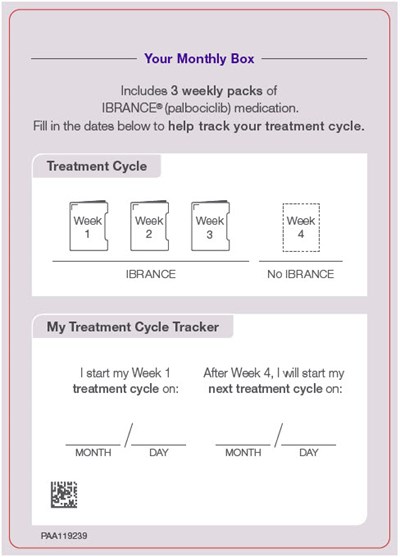
Your Monthly Box includes three weekly packs of IBRANCE medication. You can use the given tracker to fill in the dates to help track your treatment cycle. The tracker consists of three weeks, and you need to fill in the months and days for the start of the first and fourth week.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.