Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
- lbl636291117
- Structural Formula - spl image1 structure
- Figure 9 - spl image10 figure9
- Figure 10 - spl image11 figure10
- Figure 11 - spl image12 figure11
- Figure 12 - spl image13 figure12
- Figure 13 - spl image14 figure13
- Figure 1 - spl image2 figure1
- Figure 2 - spl image3 figure2
- Figure 3 - spl image4 figure3
- Figure 4 - spl image5 figure4
- Figure 5 - spl image6 figure5
- Figure 6 - spl image7 figure6
- Figure 7 - spl image8 figure7
- Figure 8 - spl image9 figure8
Product Label Images
The following 15 images provide visual information about the product associated with Donepezil Hydrochloride NDC 63629-1117 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
lbl636291117

This is a medication called Donepezil Hydrochloride, containing 23 mg that comes in a container with 30 tablets. The medication should be stored at 20° to 25°C and kept out of the reach of children. It is suggested to use a tight, light-resistant container with a child-resistant closure. The item has an NDC number of 63629-1117-1.*
Figure 9 - spl image10 figure9
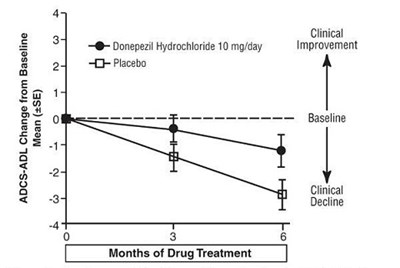
This is a table displaying the mean (with standard error) change from baseline in the ADCS-ADL for Donepezil Hydrochloride 10mg/day and Placebo groups after 3 months of drug treatment. There is also a comparison of clinical improvement vs clinical decline at baseline.*
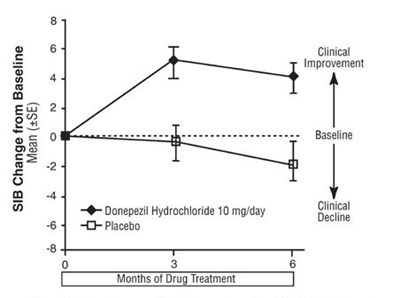
Figure 11 - spl image12 figure11
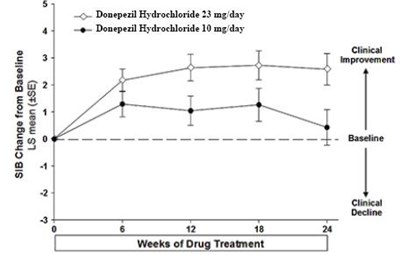
This is a record of the SIB (Severe Impairment Battery) change from baseline after drug treatment. The drug used was either Donepezil Hydrochloride at a dose of 23mg per day or Donepezil Hydrochloride at a dose of 10mg per day. The clinical improvement observed was 5 to 7 percent after a few weeks of drug treatment.*
Figure 12 - spl image13 figure12
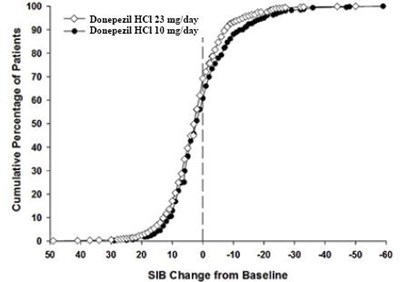
Cumulative Percentage of Patients for Donepezil HCI 23 mg/day and 10 mg/day treatment showing the change from Baseline. Data is presented in a graph format.*
Figure 13 - spl image14 figure13
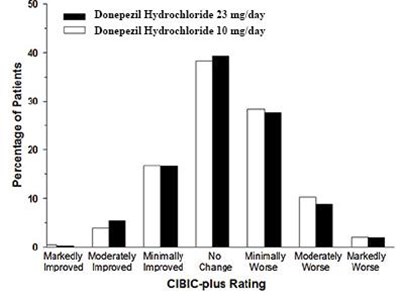
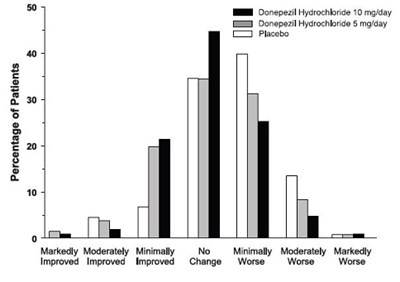
This document seems to show a table related to the percentage of patients who took Donepezil Hydrochloride in different doses, along with a rating system called CIBICplus. The table shows various levels of improvement in the patients' condition after taking the medication. However, there seems to be some errors and missing information in the text, which can make the interpretation of the table challenging.*
Figure 4 - spl image5 figure4
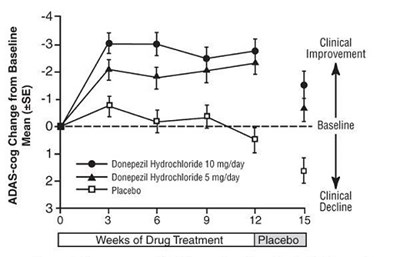
This appears to be a graph or chart showing the ADAS-cog change from baseline for three different treatments (donepezil hydrochloride 10mg/day, donepezil hydrochloride 5mg/day, and placebo). The chart shows the mean Giincal Improvement and clinical decline over the course of 15 weeks of drug treatment. There is no further information available.*
Figure 6 - spl image7 figure6
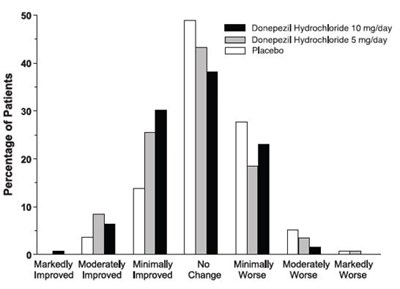
This text is a table with percentages and medication dosages. It appears to be evaluating the effectiveness of different medications. However, the table is incomplete and difficult to interpret without additional context.*
Figure 7 - spl image8 figure7
This appears to be a table showing the mean change from baseline (in some measurement) after 4 and 12 months of clinical improvement, with a comparison between patients taking Donepezil Hydrochloride 10mg/day and patients taking a placebo. The exact nature of the measurement and the units are not clear from the provided text.*
Figure 8 - spl image9 figure8
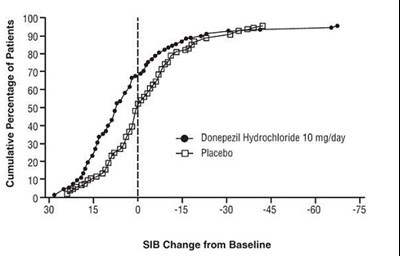
The text appears to be a chart showing the cumulative percentage of patients who took Donepezil Hydrochloride 10 mg/day and the placebo in relation to their SIB (Severe Impairment Battery) Change from Baseline. Therefore, it is a medical analysis of a study.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.