Product Images Rivastigmine Transdermal System
View Photos of Packaging, Labels & Appearance
- Label - lbl636292065
- Chemical Structure - rivastigmine 01
- Figure 1 - rivastigmine 02
- Figure 2 - rivastigmine 03
- Figure 3 - rivastigmine 04
- Figure 4 - rivastigmine 05
- Figure 5 - rivastigmine 06
- Figure 6 - rivastigmine 07
- Figure 7 - rivastigmine 08
- Figure 8 - rivastigmine 09
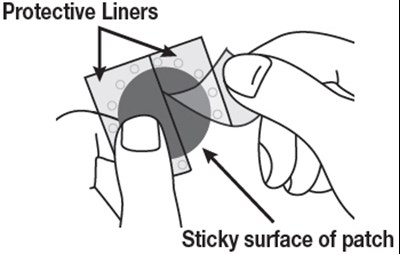
- Figure A - rivastigmine 10
- Figure B - rivastigmine 11
- Figure C - rivastigmine 12
- Figure D - rivastigmine 13
- Figure E - rivastigmine 14
- Figure F - rivastigmine 15
- Figure G - rivastigmine 16
- Figure H - rivastigmine 17
Product Label Images
The following 18 images provide visual information about the product associated with Rivastigmine Transdermal System NDC 63629-2065 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Label - lbl636292065

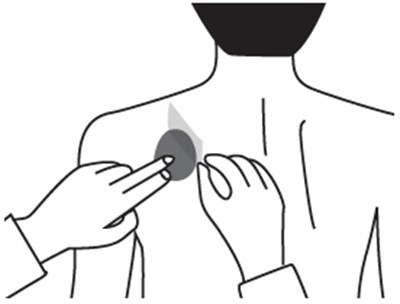
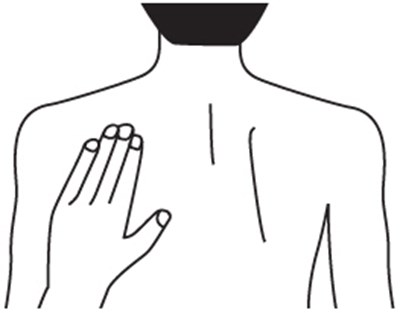
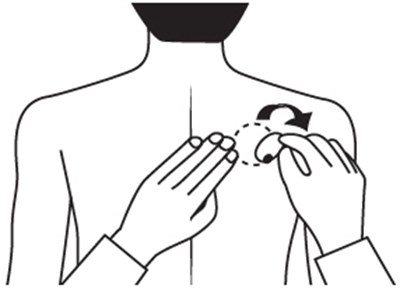
This is a description of a pharmaceutical product called Rivastigmine Transdermal System. It comes in a pack of 30 systems, with each system containing 9mg of Rivastigmine, providing a 4.6mg dosage every 24 hours. The product should be stored at a temperature between 15°C to 30°C, and wearing more than one patch at a time is prohibited. Users should apply the patch to clean and dry skin once a day and replace it every 24 hours without cutting it. The product should be kept out of reach from children as it is only for Transdermal Use Only. The National Drug Code identification number is 63629-2065-1.*
Figure 2 - rivastigmine 03

This is a table that shows the concentration (in ng/mL) of Rivastigmine, a medication used for dementia, in three different dosages (13.3 mg/24 hours, 9.5 mg/24 hours, and 4.6 mg/24 hours) taken by a total of 63 patients. There are 19 patients in the first group, 22 in the second group, and another 22 in the third group.*
Figure 3 - rivastigmine 04
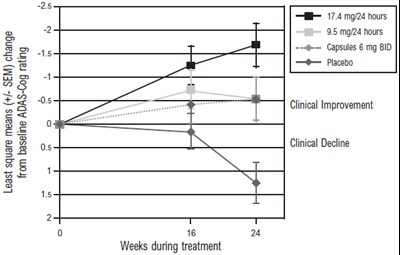
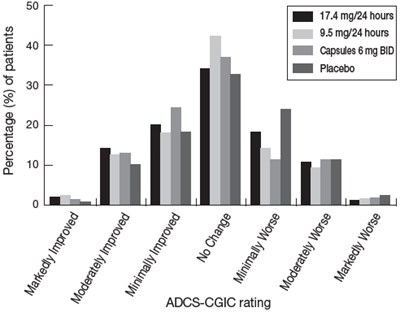
This is a table representation of the ADAS-Cog test rating. It shows the least square means with standard error mean (SEM) and the change from baseline, as well as the clinical improvement and decline at 16 weeks during the treatment. The table also displays the dosages of two different medications, Gapstles 6 mg BID, and 95 mgr24 hours, along with a placebo.*
Figure 7 - rivastigmine 08
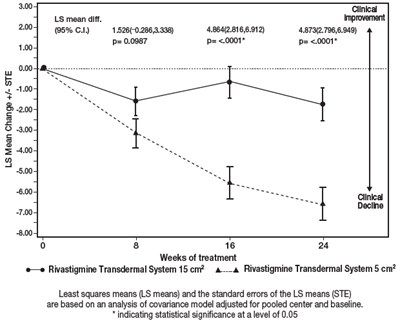
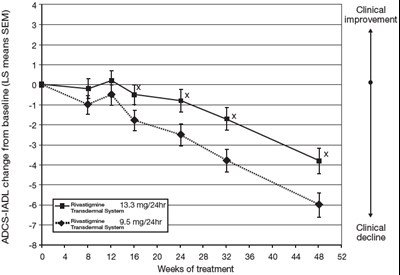
This appears to be a statistical analysis comparing the improvement from treatment with two different transdermal systems using rivastigmine. The data includes LS means and standard errors for both treatments at various time intervals (8, 16, and 24 weeks) and indicates statistical significance at a level of 0.05.*
Figure 8 - rivastigmine 09
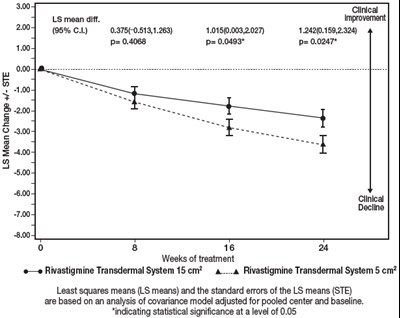
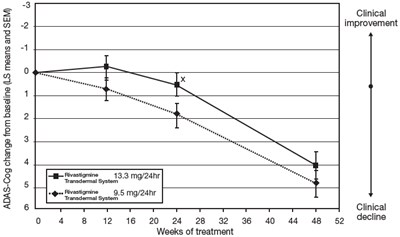
This is a description of clinical trial results for a medication called Rivastigmine Transdermal System. The trial lasted for 16 weeks and involved patients with a decline in cognitive function. Two different doses of the medication were tested and measured in least squares means (LS means) with standard errors. The results show statistical significance at a level of 0.05 for one of the doses.*
Figure B - rivastigmine 11

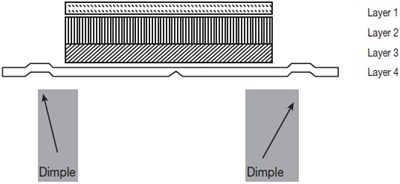
This appears to be a list of body parts with no further context or information. It is not possible to generate a useful description with this information.*
Figure H - rivastigmine 17

This text appears to be instructions for disposing of a folded patch. It is recommended to fold the sticky sides together and place the patch in the trash. Additionally, it suggests keeping the folded patch away from children and pets.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.