Product Images Rivastigmine Transdermal System
View Photos of Packaging, Labels & Appearance
- Label - lbl636292066
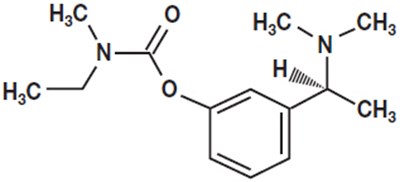
- Chemical Structure - rivastigmine 01
- Figure 1 - rivastigmine 02
- Figure 2 - rivastigmine 03
- Figure 3 - rivastigmine 04
- Figure 4 - rivastigmine 05
- Figure 5 - rivastigmine 06
- Figure 6 - rivastigmine 07
- Figure 7 - rivastigmine 08
- Figure 8 - rivastigmine 09
- Figure A - rivastigmine 10
- Figure B - rivastigmine 11
- Figure C - rivastigmine 12
- Figure D - rivastigmine 13
- Figure E - rivastigmine 14
- Figure F - rivastigmine 15
- Figure G - rivastigmine 16
- Figure H - rivastigmine 17
Product Label Images
The following 18 images provide visual information about the product associated with Rivastigmine Transdermal System NDC 63629-2066 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Label - lbl636292066

This is a description of a pharmaceutical product called NILD. It is a rivastigmine transdermal system that is used for transdermal use only. The system contains 18mg rivastigmine, providing 9.5mg rivastigmine every 24 hours. The patch should be worn for 24 hours and shouldn't be cut. It should be applied immediately to an intact skin after removal from the pouch and the location of each new patch should be changed. The patch should be stored at 25°C (77°F) with excursions permitted to 15° to 30°C (59° to 86°F). This product is available in one unit with an NDC number of 63629-2066-1 and is only for Rx use.*
Figure 2 - rivastigmine 03

This is a table providing the concentration of Rivastigmine in ng/mL, under different dosages (13.3 mg/24 hours, 9.5 mg/24 hours, 4.6mg/24 hours) and different number of patients (n=19, n=22, n=22).*
Figure 7 - rivastigmine 08

This appears to be a statistical analysis of the improvement observed in patients receiving Rivastigmine Transdermal System over a period of 8, 16, and 24 weeks of treatment. The analysis compares mean difference and standard errors of the LS means in patients receiving a 5 cm patch versus a 15 cm patch. The results indicate that patients receiving the larger patch showed a significant improvement compared to those who received the smaller patch.*
Figure 8 - rivastigmine 09

This is a clinical study evaluating the efficacy of Rivastigmine Transdermal System in improving cognitive decline over a period of 16 weeks. The study compares two dosages of the system (15 cm² and 5 cm²) and reports least squares means (LS means) and standard errors of the LS means (STE) obtained through an analysis of covariance model adjusted for pooled center and baseline. The statistical significance at a level of 0.05 is also reported.*
Figure B - rivastigmine 11
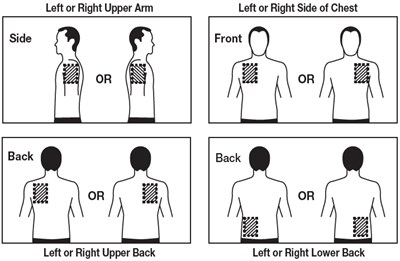
This is a list of body parts where Lenge, Z and Lk have been mentioned along with Left or Right Upper Arm, Left or Right Side of Chest, Left or Right Upper Back and Left or Right Lower Back. It is unclear what Lenge, Z and Lk indicate as the text appears to be incomplete.*
Figure H - rivastigmine 17

The text appears to be a set of instructions for disposing of a slide folded patch. The patch should be placed in the trash and the sticky sides should be folded together and placed in an empty pouch that has been saved. It also advises to keep the pouch away from children and pets.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.