Product Images Levetiracetam
View Photos of Packaging, Labels & Appearance
- e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 01
- e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 02
- e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 03
- e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 04
- e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 05
- e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 06
- e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 07
- e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 08
- e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 09
- Label Image - lbl636294137
Product Label Images
The following 10 images provide visual information about the product associated with Levetiracetam NDC 63629-4137 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 01
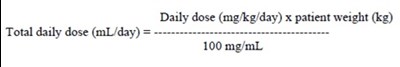
This text provides a formula for calculating the total daily dose of medication based on the patient's weight and the daily dose in milligrams per kilogram. The medication concentration is given as 100 mg/mL.*
e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 02

This appears to be a formula for calculating CLer (glomerular filtration rate) in patients. It involves multiplying the result of subtracting the patient's age (in years) from 140 by their weight (in kg) and then dividing by 72 times their serum creatinine (in mg/dL). The formula is adjusted for female patients by multiplying by 0.85. The result is then adjusted for body surface area (BSA) by dividing by the subject's BSA (in square meters, m²) and multiplying by 1.73.*
e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 05
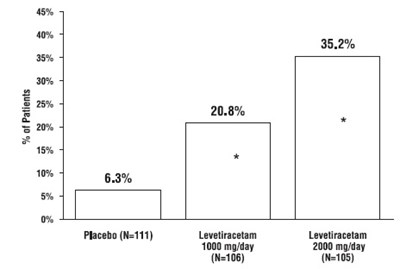
This appears to be a chart showing the percentage of patients in a study who experienced certain outcomes while taking either a placebo or different dosages of a medication called Levetiracetam. The outcomes are not specified in the given text.*
e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 07
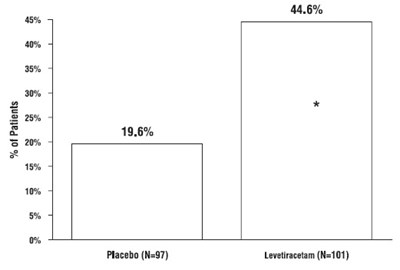
This document appears to be a graphical presentation of patient data that compares the effectiveness of a placebo drug against Levetiracetam. The chart seems to show the percentage of patients in each category of response to the respective drugs, but there isn't enough text to determine what those categories may be.*
e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 08

This appears to be a table with two columns showing the percentage of patients in a study who were given either a placebo or a medication called Levetiracetam. The first column shows that 19.6% of the patients in the study were given a placebo (a substance with no therapeutic effect), while the second column shows that 43.1% of the patients in the study were given Levetiracetam. The "N" followed by a number in parentheses is likely showing the number of patients in each group.*
e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 09
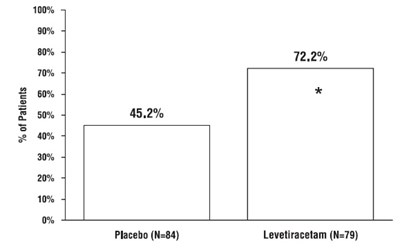
This is a chart displaying the percentage of patients in a study. The study involved a placebo group of 84 people and a levetiracetam group of 79 people. The percentages shown relate to the efficacy or side effects of the treatments. However, there is not enough data available to determine which category each percentage refers to.*
Label Image - lbl636294137

This appears to be the packaging information for a drug called Keppra 500mg Tablet, manufactured by Lupin Pharmaceuticals, Inc. The package contains 90 tablets and the expiration date is listed as MM/YY. The National Drug Code (NDC) is 6362941371. It is recommended to keep all drugs out of reach of children and store at room temperature between 20-25°C (68°-77°F).*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


