Product Images Alfuzosin Hydrochloride
View Photos of Packaging, Labels & Appearance
- ef35abcf e0b6 4551 84cf a689ad11e6cd 01
- ef35abcf e0b6 4551 84cf a689ad11e6cd 02
- ef35abcf e0b6 4551 84cf a689ad11e6cd 03
- ef35abcf e0b6 4551 84cf a689ad11e6cd 04
- ef35abcf e0b6 4551 84cf a689ad11e6cd 05
- ef35abcf e0b6 4551 84cf a689ad11e6cd 06
- ef35abcf e0b6 4551 84cf a689ad11e6cd 07
- ef35abcf e0b6 4551 84cf a689ad11e6cd 08
- ef35abcf e0b6 4551 84cf a689ad11e6cd 09
- ef35abcf e0b6 4551 84cf a689ad11e6cd 10
- Label Image - lbl636297907
Product Label Images
The following 11 images provide visual information about the product associated with Alfuzosin Hydrochloride NDC 63629-7907 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
ef35abcf e0b6 4551 84cf a689ad11e6cd 03
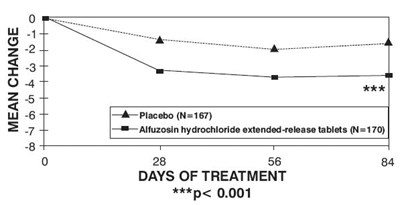
This appears to be a graph or table showing the mean change in some measure (not specified) over the course of 28, 56, and 84 days of treatment, comparing a placebo group (N=167) to a group taking a medication called Altuzosin hydrochloride extended-release tablets (N=170). The asterisks next to the "p<0.001" suggest that the difference between the two groups is statistically significant. However, without more information about what is actually being measured, it is difficult to provide a more specific description.*
ef35abcf e0b6 4551 84cf a689ad11e6cd 04
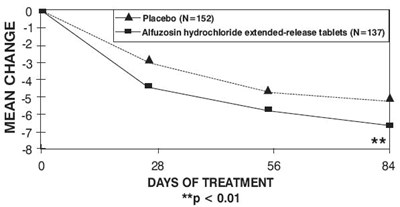
The text describes a study, which involved two groups of people. One group (N=152) received placebo, while the other group (N=137) received Alfuzosin hydrochloride extended-release tablets. The study lasted for 28, 56, and 84 days of treatment. The asterisks indicate that the results were statistically significant (p<0.01). The text does not provide further details on the study or its findings.*
ef35abcf e0b6 4551 84cf a689ad11e6cd 05
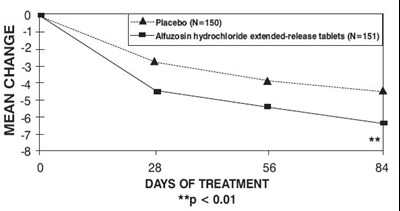
This is a chart showing the mean change over days of treatment for a placebo group and a group treated with Altuzosin hydrochloride extended-release tablets. The chart indicates significant improvement in the group treated with Altuzosin hydrochloride.*
ef35abcf e0b6 4551 84cf a689ad11e6cd 06
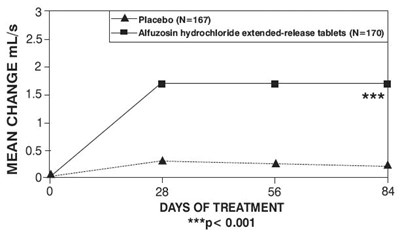
The text describes the mean change in mL/s over a period of 56 to 84 days of treatment using a placebo and alfuzosin hydrochloride extended-release tablets, with a significant difference between the two treatments.*
ef35abcf e0b6 4551 84cf a689ad11e6cd 07
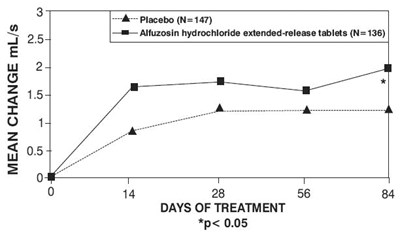
This is a graph showing the mean change in mL/s in patients receiving placebo (N=147) and alfuzosin hydrochloride extended-release tablets (N=136) over a period of 84 days. The graph indicates that alfuzosin hydrochloride extended-release tablets were more effective in improving mL/s as compared to placebo. The asterisk indicates statistical significance with p<0.05.*
Label Image - lbl636297907

This medication is called Alfuzosin Hydrochloride, which comes in the form of white round tablets marked with U;155. Each tablet is 10mg and may cause dizziness. The tablets are packaged by Bryant Ranch Prepack Burbank and should be stored at room temperature between 20°-25°C (68°-77°F). The expiration date for this batch of tablets is not mentioned. This medicine is a generic version of Uroxatral 10mg tablet and is sold under the brand Liberprost. It is recommended to keep all medications out of reach of children.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.