Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
- 01 - 30406488 figure 01
- figure2 - 30406488 figure 02
- figure-03 - 30406488 figure 03
- figure-04 - 30406488 figure 04
- figure-05 - 30406488 figure 05
- figure-06 - 30406488 figure 06
- figure-07 - 30406488 figure 07
- 08 - 30406488 figure 08
- 09 - 30406488 figure 09
- 10 - 30406488 figure 10
- 11 - 30406488 figure 11
- 12 - 30406488 figure 12
- 13 - 30406488 figure 13
- 14 - 30406488 figure 14
- 15 - 30406488 figure 15
- Label - lbl636298483
Product Label Images
The following 16 images provide visual information about the product associated with Donepezil Hydrochloride NDC 63629-8483 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure2 - 30406488 figure 02
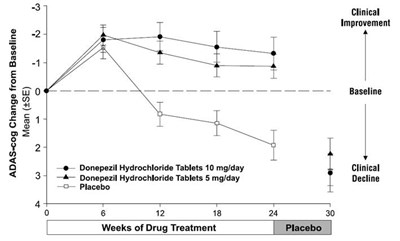
This is a chart showing the change from baseline in ADAS-cog score after weeks of drug treatment with Donepezil Hydrochloride Tablets 10 mg/day, Donepezil Hydrochloride Tablets 5 mg/day, and Placebo. The chart shows mean values with SE for each treatment group at weeks 6, 12, 18, and 24. The chart also indicates clinical improvement or decline for the placebo group.*
figure-03 - 30406488 figure 03
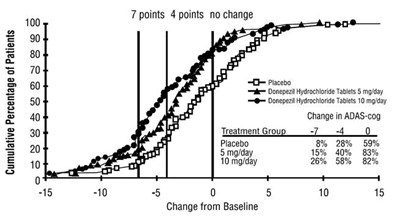
This is a graph that displays the percentage of patients who demonstrated improvement, no change, or decline in their Alzheimer's Disease Assessment Scale-Cognitive Subscale (ADAS-cog) scores over the treatment course. The x-axis shows the change from baseline ADAS-cog scores by 15-point increments while the y-axis shows the percentage of patients. The graph compares the outcomes of patients treated with a placebo against those treated with 5mg/day and 10mg/day of donepezil hydrochloride tablets. Patients treated with donepezil hydrochloride tablets demonstrated a higher percentage of improvement and a lower percentage of decline compared to the placebo group.*
figure-04 - 30406488 figure 04

This is data related to the percentage of patients on various doses of Donepezil Hydrochloride Tablets (10 mg/day and 5 mg/day) as well as placebo. The graph shows the CIBIC-plus rating with seven different categories ranging from markedly improved, moderately improved, minimally improved, no change, and worse, and even markedly worse. Further information is not available.*
figure-05 - 30406488 figure 05
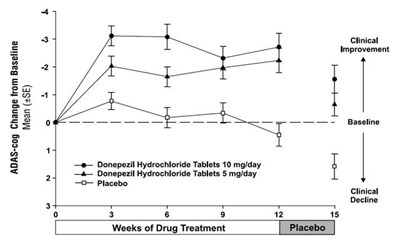
The text describes the results of a clinical study measuring the change from baseline in the ADAS-cog score for patients receiving different treatments over a period of 12 weeks. The treatments evaluated were donepezil hydrochloride tablets at doses of 5mg/day and 10mg/day, and a placebo. The table displays the mean change in ADAS-cog score (+SE) and clinical improvement for each treatment at baseline and at weeks 0, 3, 6, 9, and 12.*
08 - 30406488 figure 08

This text provides a comparison of the mean change from baseline in SIB (Severe Impairment Battery) scores between the treatment group receiving Donepezil Hydrochloride Tablets 10 mg/day and the placebo group over a period of three months. It also indicates that clinical improvement was measured at baseline but does not provide any further information.*
10 - 30406488 figure 10

This is a graph representing the change from baseline in ADCS-ADL score. The mean score with standard error is presented for placebo and Donepezil Hydrochloride Tablets 10 mg/day over a period of 3 months of drug treatment. The graph shows that there is clinical improvement over the baseline with Donepezil Hydrochloride Tablets 10 mg/day, while the placebo shows a clinical decline.*
11 - 30406488 figure 11
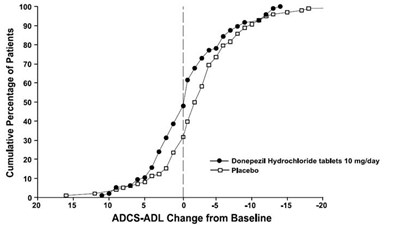
This is a graph that shows the change from baseline in ADCS-ADL for patients treated with Donepezil Hydrochloride tablets at a dose of 10mg/day compared to patients who received a placebo. The graph has various numerical values labeled on the horizontal axis ranging from -20 to 20, which represent ADCS-ADL score changes in patients. The vertical axis is not labeled.*
13 - 30406488 figure 13
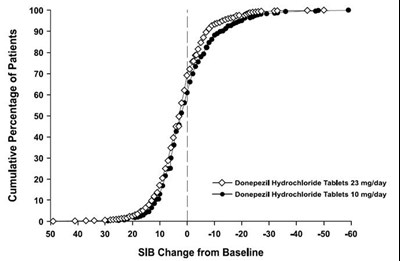
The image appears to be a graph displaying the cumulative percentage of patients and the SIB change from baseline with two different doses of Donepezil Hydrochloride Tablets (10 mg/day and 23 mg/day). The x-axis shows the SIB Change from Baseline (with ticks at 0, 10, 20, 30, 40, 50, and 60), and the y-axis shows the cumulative percentage of patients (with ticks at 0, 50, and 100). The line for the 23 mg/day dose of Donepezil is shown in solid circles (labeled with 'O'), while the line for the 10 mg/day dose is shown in 'e'. The horizontal line at the top of the graph represents 100% of patients, while the dashed line at the half-way point of the graph represents 50% of patients.*
14 - 30406488 figure 14
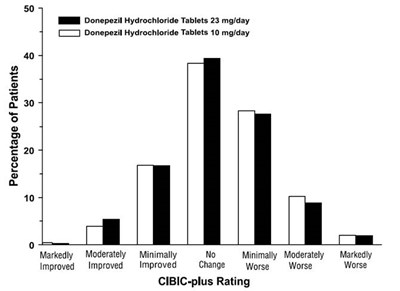
This appears to be a chart comparing the effectiveness of Donepezil Hydrochloride Tablets of different dosages (10mg/day and 23mg/day) on patients. The chart shows the percentage of patients who experienced marked improvement, moderate improvement, minimal improvement, no improvement, or worsening of their conditions based on the CIBIC-plus rating.*
Label - lbl636298483

Donepezil Hydrochloride is a useful medication in the management of dementia. Each tablet contains USP 23 mg of Donepezil Hydrochloride. Store the tablets in a dry place at a temperature of 20° to 25°C (68° to 77°F) to retain their efficacy. It is essential to dispense them in a light-resistant, sealed container to prevent degradation. Do not purchase the tablets if the seal over the bottle is missing or broken. The tablets are available in a pack of 30 tablets and a prescription from a licensed physician is required to obtain them.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.





