Product Images Fluticasone Propionate And Salmeterol
View Photos of Packaging, Labels & Appearance
- FP chem structure - fluticasone propionate salmeterol inhalation powde 1
- Figure 8 - fluticasone propionate salmeterol inhalation powde 10
- Teva Logo - fluticasone propionate salmeterol inhalation powde 11
- Teva Logo - fluticasone propionate salmeterol inhalation powde 12
- Figurr A - fluticasone propionate salmeterol inhalation powde 13
- Figure B - fluticasone propionate salmeterol inhalation powde 14
- Figure C - fluticasone propionate salmeterol inhalation powde 15
- Figure C - fluticasone propionate salmeterol inhalation powde 16
- Breathe out image - fluticasone propionate salmeterol inhalation powde 17
- Figure F - fluticasone propionate salmeterol inhalation powde 18
- Figure G - fluticasone propionate salmeterol inhalation powde 19
- Salmeterol chem structure - fluticasone propionate salmeterol inhalation powde 2
- Figure H - fluticasone propionate salmeterol inhalation powde 20
- Teva Logo - fluticasone propionate salmeterol inhalation powde 21
- Figure 1 - fluticasone propionate salmeterol inhalation powde 3
- Figure 2 - fluticasone propionate salmeterol inhalation powde 4
- Figure 3 - fluticasone propionate salmeterol inhalation powde 5
- Figure 4 - fluticasone propionate salmeterol inhalation powde 6
- Figure 5 - fluticasone propionate salmeterol inhalation powde 7
- Figure 6 - fluticasone propionate salmeterol inhalation powde 8
- Figure 7 - fluticasone propionate salmeterol inhalation powde 9
- Label - lbl636298811
Product Label Images
The following 22 images provide visual information about the product associated with Fluticasone Propionate And Salmeterol NDC 63629-8811 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
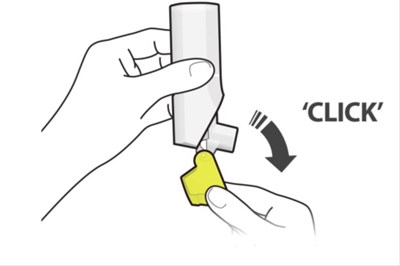
Figurr A - fluticasone propionate salmeterol inhalation powde 13

The text describes a medical inhaler device that has a dose counter, a mouthpiece, and a yellow cap. The name of the device is not available.*
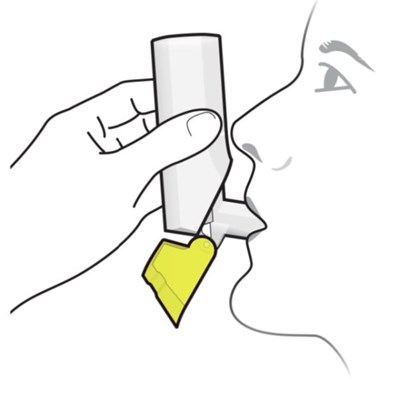
Figure B - fluticasone propionate salmeterol inhalation powde 14

This text is providing simple information regarding the number of doses available in an inhaler, whether it's full or empty. It mentions that the full inhaler contains 60 doses whereas there are 0 doses available in an empty inhaler.*
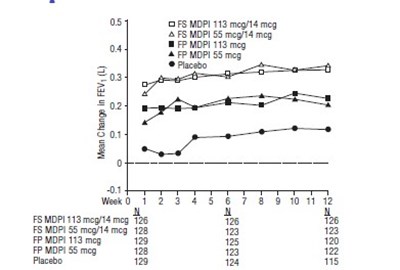
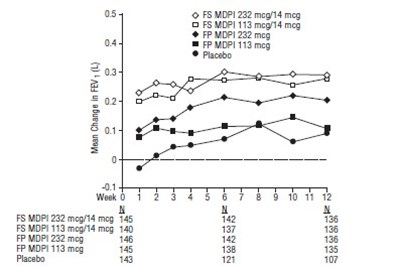
Figure 4 - fluticasone propionate salmeterol inhalation powde 6

This text refers to the results of a study conducted on various types of medications and a placebo. The medications mentioned include O FS MDPI 113 mcg/14 mcg, 05 & FS MDPI 55 mcg/14 mcg, FP MDPI 113 mcg, and A FP MDP15S mcg, with sample sizes of 61, 56, 72, and 63 respectively. The study measured the mean change in FEV (forced expiratory volume) in liters over the course of one day, with baseline measurements taken at hour 0 and measurements taken at hours 1, 2, 3, 4, 5, 6, 7, 9, 10, and 11. The results for placebo are also included with a sample size of 60.*
Label - lbl636298811

Fluticasone Propionate and Salmeterol Inhalation Powder is a prescription drug. It is a combination of Fluticasone proprionate and Salmeterol base. Each metered dose of the inhaler contains 55 mcg of Fluticasone proprionate and 14 mcg of Salmeterol base. It should be used for oral inhalation only. The drug is available in a pack of 60 metered inhalations (0.45 g). The inhaler should not be washed or put in water. It should be stored in a controlled room temperature between 15°C and 25°C (59°F and 77°F) and should be protected from extreme heat, cold, or humidity.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.