Product Images Buprenorphine And Naloxone
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Buprenorphine And Naloxone NDC 63629-9482 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
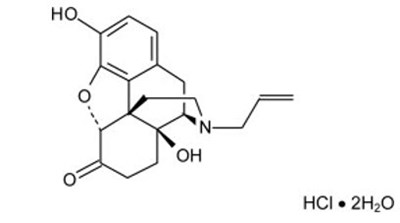
Chemical Structure - buprenorphine 01
HCI stands for Human-Computer Interaction. This field is focused on the design, evaluation, and implementation of interactive computer systems for human use. HCI aims to create user-friendly and efficient digital interfaces by considering psychological, social, and cultural factors. It incorporates principles from computer science, design, psychology, and anthropology to improve user experience and productivity. Examples of HCI include website design, mobile apps, video games, and virtual reality systems.*
Label - lbl636299482

This is a pharmaceutical product description for Buprenorphine and Naloxone Sublingual Tablets, USP with a strength of 2 mg/0.5 mg. The tablets are to be stored at a temperature range of 20° to 25°C and should be kept out of reach of children. The product has potential harmful effects, causing children accidentally taking it to need emergency medical care. The description includes the content of the tablets, which contain 2.16mg buprenorphine hydrochloride, equivalent to 2 mg buprenorphine base, and 0.61 mg naloxone hydrochloride dihydrate, equivalent to 0.5 mg naloxone base, per sublingual tablet. The document lists a National Drug Code of 63629-9482-01 and provides prescribing information for pharmacists.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.