Product Images Lansoprazole
View Photos of Packaging, Labels & Appearance
Product Label Images
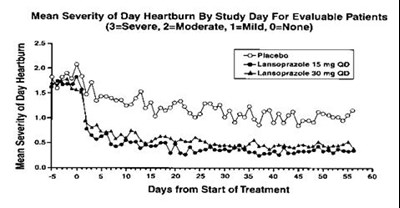
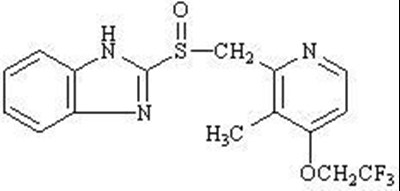
The following 6 images provide visual information about the product associated with Lansoprazole NDC 63739-555 by Mckesson Corporation Dba Sky Packaging, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
30mg 10x10

This is a description of Lansoprazole delayed-release capsules, USP. The capsules come in 30g dose with enteric coating and are produced in India. The label also includes a lot number and a product code.*
Label Image - lbl63739555

This is a medication label for Lansoprazole Delayed-Release Capsules, USP with a specific National Drug Code (NDC) of 63730-565-41. The capsules are indicated for prescription use only and contain 30 capsules. The label also contains a statement advising to consult the medication guide and pharmacist for usage instructions. The remaining text is not legible due to errors.*
Label Image - lbl63739574

This is a description of a medication called Lansoprazole. It comes in the form of delayed-release capsules and is available in a strength of 15 mg. The medication is packaged in blister packs, with 10 blisters of 10 capsules each, totalling 100 capsules per package. The medication is identified by a National Drug Code (NDC) of 63739-574-10 and a Trade Name (TN) of 0. The meaning of the other letters and numbers, such as "UD," "I L," and "rasky 555," is not clear and may not be relevant to the medication description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.