Product Images Propafenone Hydrochloride
View Photos of Packaging, Labels & Appearance
- Image - 74c908e9 3577 40a8 81e0 e6012106181f 01
- Image - 74c908e9 3577 40a8 81e0 e6012106181f 02
- Image - 74c908e9 3577 40a8 81e0 e6012106181f 03
- Image - 74c908e9 3577 40a8 81e0 e6012106181f 04
- Image - 74c908e9 3577 40a8 81e0 e6012106181f 05
- Image - 74c908e9 3577 40a8 81e0 e6012106181f 06
- Image - 74c908e9 3577 40a8 81e0 e6012106181f 07
- Image - 74c908e9 3577 40a8 81e0 e6012106181f 08
- Image - 74c908e9 3577 40a8 81e0 e6012106181f 09
- Image - 74c908e9 3577 40a8 81e0 e6012106181f 10
- Image - 74c908e9 3577 40a8 81e0 e6012106181f 11
Product Label Images
The following 11 images provide visual information about the product associated with Propafenone Hydrochloride NDC 64380-184 by Strides Pharma Science Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image - 74c908e9 3577 40a8 81e0 e6012106181f 02
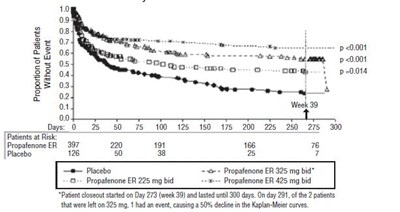
This is a graph representing the proportion of patients without an event over time. The patients were treated with either Propafenone ER or placebo. There were a total of 307 patients in the study, with varying dosages of Propafenone ER. One patient had an event on day 231 which resulted in a 50% decline in the Kaplan-Meier curves. The study lasted for 300 days.*
Image - 74c908e9 3577 40a8 81e0 e6012106181f 03
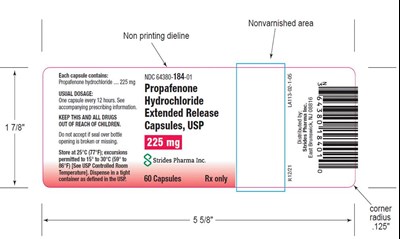
This is a medication label with instructions for use and dosage information. The medication is available in capsule form, with a recommended dosage of one capsule every 12 hours. The label warns against accepting a bottle with a broken seal and emphasizes the need to keep the drug out of the reach of children. The label also provides storage instructions and contact information for the pharmaceutical company that distributes the medication. Additionally, there is an image of a non-printing dieline with a non-varnished area and a corner radius of 125 inches.*
Image - 74c908e9 3577 40a8 81e0 e6012106181f 04
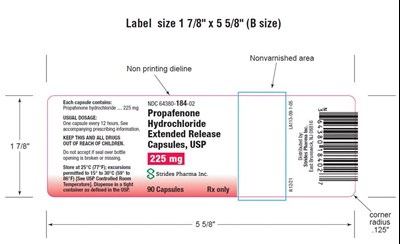
This is a label size description with dimensions of 17/8" x 5 5/8" (B size). There is also a non-printing dieline present. The text includes information about a capsule containing Froptenon yiucnone and its recommended dosage. Additionally, it provides information about the storage temperature and cautions the reader to keep the capsules out of reach of children. Lastly, it notes the manufacturer and includes some dimensions for the label.*
Image - 74c908e9 3577 40a8 81e0 e6012106181f 05
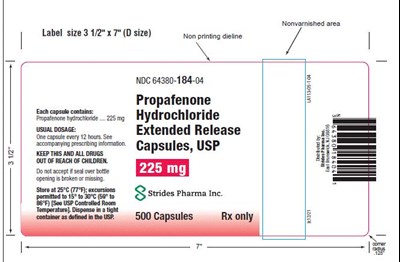
This is a label for Propafenone Hydrochloride Extended Release capsules. The label size is 3 12 x 7" (D size) and has a non-printing area. The capsules should be taken every 12 hours and contain 225mg. The manufacturer is Sennmomngeren S strides Pharma Inc. The container has 500 capsules and is available only by prescription.*
Image - 74c908e9 3577 40a8 81e0 e6012106181f 06
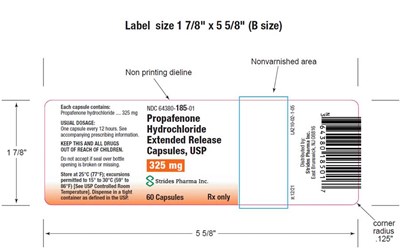
Label size 17/8" x5 5/8" (B size). Nonvarnished area. This document appears to be a prescription drug label with instructions for usage and storage indicated.*
Image - 74c908e9 3577 40a8 81e0 e6012106181f 07
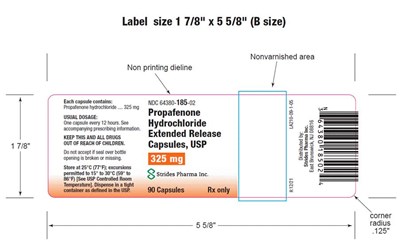
This text is describing a label for Propafenone hydrochloride Extended Release capsules. It includes details such as the size of the non-printed dieline, non-vanished area, and dosage instructions. The capsules contain 325 mg of Propafenone hydrochloride, and the usual dosage is one capsule every 12 hours. The label also includes information on storing and dispensing the capsules, as well as the NDC and manufacturer information. There are also measurements provided for the size of the label corners.*
Image - 74c908e9 3577 40a8 81e0 e6012106181f 08
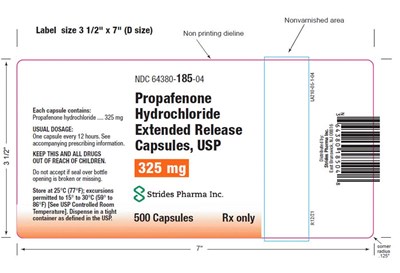
The text contains information about a label size of 3.5" x 7", non-printing dieline, non-varnished area, NDC code 64380-185-04 E, and Propafenone hydrochloride 325mg extended-release capsules. It also mentions typical dosage for the medicine and storage instructions. However, the text is not well-structured and some characters are missing, so the overall comprehension of the content is affected.*
Image - 74c908e9 3577 40a8 81e0 e6012106181f 09
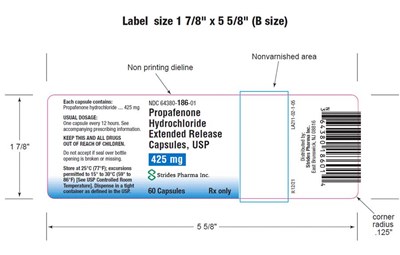
This label size is 1 7/8" x 5 5/8" and is classified as B size. There is a nonvarnished area and a non-printing dieline included. The label contains information about ropafenone hydrochloride extended-release capsules. The recommended storage temperature for the capsules is 25°C (77°F) and excursions are permitted to 15-30°C (59-86°F). This label also includes a measurement of 125" radius and dimensions of 5 5/8" for the length along with a corner.*
Image - 74c908e9 3577 40a8 81e0 e6012106181f 10
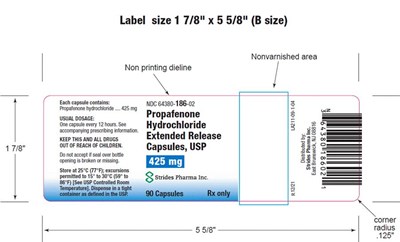
Label size is 1 7/8" x 5 5/8" (B size). The label contains instructions and information regarding the dosage of a drug and its accompanying prescribing format. It also cautions against accepting damaged or broken capsules and recommends storage below 25°C. The label is produced by Strides Pharma Inc.*
Image - 74c908e9 3577 40a8 81e0 e6012106181f 11

This is a drug label for Propafenone Hydrochloride Extended Release Capsules, with a size of 3 1/2" x 7" and containing 425 mg. Its usual dosage is one capsule every 12 hours. The label also includes storage instructions and a warning to keep it out of reach of children. The label is distributed by Strides Pharma and has an NDC number of 64380-186-04. Additional information regarding the file could not be read.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
