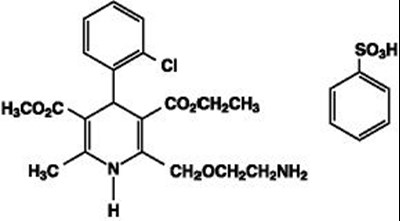
Product Images Amlodipine And Valsartan
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Amlodipine And Valsartan NDC 64380-216 by Strides Pharma Science Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1: Probability of Achieving Systolic Blood Pressure <140 mmHg at Week 8 - 00cf9c79 c9d7 42a7 9208 404fcee14f11 11

The text provided appears to be a mixture of prescription drug information with some numerical readings. It is not possible to generate a useful description with this text.*
Figure 2: Probability of Achieving Diastolic Blood Pressure <90 mmHg at Week 8 - 00cf9c79 c9d7 42a7 9208 404fcee14f11 12

The text indicates blood pressure measurements with a baseline DBP (diastolic blood pressure) of 80 mmHg or less. There is also mention of possible medications, including Amlodipine, Losartan, and a 5% pacemaker, among others. However, the context and purpose of this information are not clear.*
Figure 3: Probability of Achieving Systolic Blood Pressure <130 mmHg at Week 8 - 00cf9c79 c9d7 42a7 9208 404fcee14f11 13

This text is not available for evaluation as it does not provide sufficient information to generate a coherent description.*
Figure 4: Probability of Achieving Diastolic Blood Pressure <80 mmHg at Week 8 - 00cf9c79 c9d7 42a7 9208 404fcee14f11 14

This text seems to be a blood pressure measurement, indicating that the diastolic blood pressure (DBP) is less than 80 millimeters of mercury (mmHg). It also includes a baseline measurement of DBP in mmHg. It's possible that this information comes from a medical or scientific record.*
Container Label 5mg/160mg - 00cf9c79 c9d7 42a7 9208 404fcee14f11 17

This document contains multiple pieces of information that are not related to each other. It starts with a "Non printing dieline" followed by an empty space and "Nonvarnished area". Then, there are some numbers and symbols like "112", NDC 64380-214-01 2\", Amlodipine besylate USP, etc. The text mentions usual dosage and package insert details but not for what drug. It recommends keeping the tablets out of reach of children. The document suggests storing the tablets at a temperature range of 25°C to 30°C and protecting them from moisture. The final line mentions something about a radius of 0.125 and a corner of 45/8".*
Image - 00cf9c79 c9d7 42a7 9208 404fcee14f11 18

This is a description of a medication called Amlodipine and Valsartan tablets. Each tablet contains 10mg of Amlodipine besylate USP and 160mg of Valsartan. The usual dosage information can be found in the package insert. It is important to keep this medication and all drugs out of reach of children and store it at a temperature between 15°C to 30°C. The NDC number is 64380-215-01 and it is distributed by Strides Pharma Inc. The packaging also includes a corner radius measurement.*
Image - 00cf9c79 c9d7 42a7 9208 404fcee14f11 19

This appears to be a combination of medical dosage information and technical printing specifications. The text mentions a tablet containing a combination of Acipneteoyte and Amlodipine, with Valsarta also included. The dosage and storage information is listed, along with the manufacturer (Strides Pharma Inc). The text also includes technical details about printing including a nonvarnished area, a dieline, and a corner radius measurement.*
Container Label 10mg/320mg - 00cf9c79 c9d7 42a7 9208 404fcee14f11 20

Each tablet contains Amlodipine and Valsartan. The usual dosage is mentioned in the package insert. It is important to keep the tablets out of the reach of children and store them at the recommended temperature. There is also a nonvarnished area and non-printing dieline mentioned which may be related to the tablet packaging.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.