Product Images Gabapentin
View Photos of Packaging, Labels & Appearance
- Cockcroft and Gault Equation - f0697d39 1473 4df8 b955 8ad587c02c5f 01
- Image - f0697d39 1473 4df8 b955 8ad587c02c5f 02
- Figure 1. Weekly Mean Pain Scores (Observed Cases in ITT Population): Study 1 - f0697d39 1473 4df8 b955 8ad587c02c5f 03
- Figure 2. Weekly Mean Pain Scores (Observed Cases in ITT Population): Study 2 - f0697d39 1473 4df8 b955 8ad587c02c5f 04
- Figure 3. Proportion of Responders (patients with ≥ 50% reduction in pain score) at Endpoint: Controlled PHN Studies - f0697d39 1473 4df8 b955 8ad587c02c5f 05
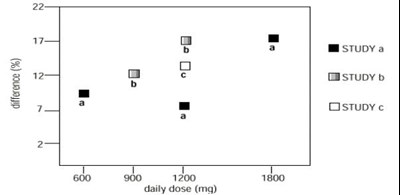
- Figure 4. Responder Rate in Patients Receiving Gabapentin Expressed as a Difference from Placebo by Dose and Study: Adjunctive Therapy Studies in Patients ≥ 12 Years of Age with Partial Seizures - f0697d39 1473 4df8 b955 8ad587c02c5f 06
- Gabapentin Capsules 100mg - 100S - HDPE Bottle Label - f0697d39 1473 4df8 b955 8ad587c02c5f 07
- Gabapentin Capsules 300mg - 100S - HDPE Bottle Label - f0697d39 1473 4df8 b955 8ad587c02c5f 08
- Gabapentin Capsules 400mg - 100S - HDPE Bottle Label - f0697d39 1473 4df8 b955 8ad587c02c5f 09
Product Label Images
The following 9 images provide visual information about the product associated with Gabapentin NDC 64380-867 by Strides Pharma Science Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Cockcroft and Gault Equation - f0697d39 1473 4df8 b955 8ad587c02c5f 01
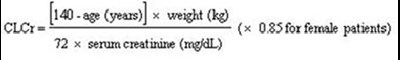
This appears to be a formula used to calculate a patient's estimated creatinine clearance (CLCr). It takes into account the patient's age, weight, and serum creatinine levels. However, without more information or context, it is not possible to provide a more detailed description of what this formula is used for or how it is applied in a medical setting.*
Figure 1. Weekly Mean Pain Scores (Observed Cases in ITT Population): Study 1 - f0697d39 1473 4df8 b955 8ad587c02c5f 03
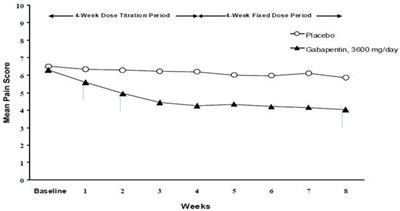
Figure 2. Weekly Mean Pain Scores (Observed Cases in ITT Population): Study 2 - f0697d39 1473 4df8 b955 8ad587c02c5f 04
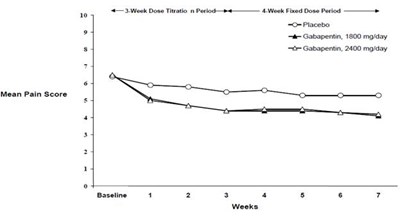
This text appears to be a chart or table with three treatment groups (placebo, 1800 mg/day of gabapentin, and 2400 mg/day of gabapentin) and a pain score measured over seven weeks. The title of the chart is "Mean Pain Score" and it is unclear what condition or type of pain this refers to. Without additional information or context, it is difficult to provide a more detailed description.*
Figure 3. Proportion of Responders (patients with ≥ 50% reduction in pain score) at Endpoint: Controlled PHN Studies - f0697d39 1473 4df8 b955 8ad587c02c5f 05
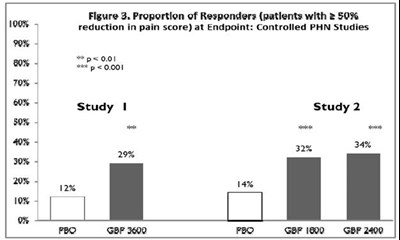
This is a chart showing the proportion of responders, patients with a 50% to 100% reduction in pain score, at the endpoint in controlled studies of Postherpetic neuralgia (PHN). The chart includes data from Study 1 and Study 2. There are different doses studied, FBO, GBF 3600, GBF 1800, and GBF 2400. The percentage of responders varies between 12% and 34%. The asterisk above the graph indicates that p<0.001.*
Figure 4. Responder Rate in Patients Receiving Gabapentin Expressed as a Difference from Placebo by Dose and Study: Adjunctive Therapy Studies in Patients ≥ 12 Years of Age with Partial Seizures - f0697d39 1473 4df8 b955 8ad587c02c5f 06
Gabapentin Capsules 100mg - 100S - HDPE Bottle Label - f0697d39 1473 4df8 b955 8ad587c02c5f 07

Gabapentin Capsules is a prescription drug containing 100mg of Gabapentin USP. The medication guide should be dispensed to each patient by the pharmacist. The usual dosage is not available, and the complete prescribing information can be found in the accompanying teralure. The capsules should be stored at a temperature between 15°C to 30°C (59°F 10 86°F) and out of reach of children. The container should be tightly closed, and the contents should be dispensed in a tight light-resistant container with a child-resistant closure, as required. The product is manufactured by Strides Pharma Science Lid in India and distributed by Strides Pharma Inc in East Brunswick, NJ. NDC 64380-867-07.*
Gabapentin Capsules 300mg - 100S - HDPE Bottle Label - f0697d39 1473 4df8 b955 8ad587c02c5f 08

This is a product label for Gabapentin capsules. The capsules contain 300mg of Gabapentin and are intended for oral use. The medication should be stored at a temperature of 50°F to 86°F and kept out of reach of children. The label also includes information on the manufacturer, dosage instructions, and a warning to consult the medication guide before use.*
Gabapentin Capsules 400mg - 100S - HDPE Bottle Label - f0697d39 1473 4df8 b955 8ad587c02c5f 09

This appears to be a label for Gabapentin capsules in the USP form. It provides information on the recommended dosage, the container for storage, and the content of the capsules. The manufacturer is identified as Stides Pharma Science Ltd. However, there might be some typographical errors in the text as it is obtained via .*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.