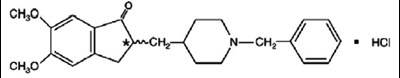
Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 20 images provide visual information about the product associated with Donepezil Hydrochloride NDC 64380-908 by Strides Pharma Science Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure 2 - eb0ec3ab 9077 4d91 a965 4b8ed556951f 03

The text describes a chart showing the cumulative percentage of patients in different treatment groups based on change in ADAS-cog score from baseline. The treatment groups include Donepezil hydrochloride 10mg/day, Donepezil hydrochloride 5mg/day, and Placebo. The chart also shows percentages of patients with 7, 4, or no change in ADAS-cog score.*
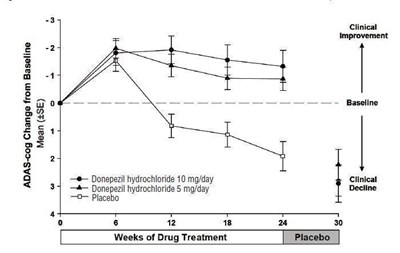
figure 4 - eb0ec3ab 9077 4d91 a965 4b8ed556951f 05

The text represents a chart displaying the changes in ADAS-cog scores of patients who were given Donepezil hydrochloride 10 mg/day, Donepezil hydrochloride 5 mg/day or Placebo at different weeks of drug treatment. The chart shows that patients given Donepezil hydrochloride 10 mg/day and Donepezil hydrochloride 5 mg/day show a clinical improvement while placebo patients show clinical decline. The text is not complete and does not provide sufficient details.*
figure 5 - eb0ec3ab 9077 4d91 a965 4b8ed556951f 06

The text appears to be a table showing the "Cumulative Percentage of Patients" in a treatment group compared to a placebo group with varying doses of "Donepezil hydrochloride". The table also includes "Change in ADAS-cog" and "Change from Baseline" percentages. The text is not clear enough to provide further information.*
figue 6 - eb0ec3ab 9077 4d91 a965 4b8ed556951f 07

This appears to be a chart showing the percentage of patients who either improved or got worse while taking Donepezil hydrochloride 10 mg/day, Donepezil hydrochloride 5 mg/day or Placebo. The chart seems to be based on the CIBIC-plus Rating of patients. However, there is no clear indication of what the chart is for or what the CIBIC-plus Rating represents.*
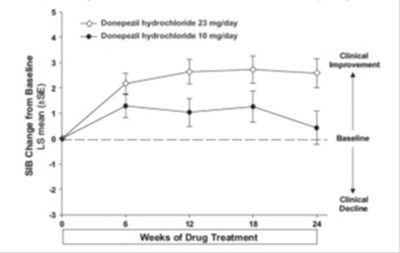
figure 7 - eb0ec3ab 9077 4d91 a965 4b8ed556951f 08

The text describes a table with the mean (+SE) change from baseline in SIB (Severe Impairment Battery) scores after 3 months of drug treatment with 10 mg/day of donepezil hydrochloride and placebo. The table also shows clinical decline in the scores.*
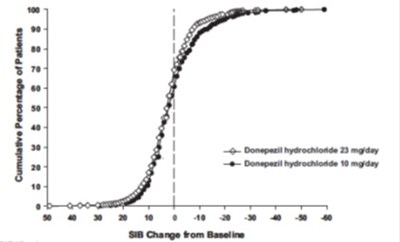
figure 8 - eb0ec3ab 9077 4d91 a965 4b8ed556951f 09
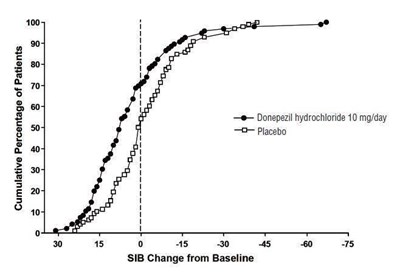
The text describes a graph that shows the cumulative percentage of patients who took either Donepezil hydrochloride or Placebo for a certain period of time. The graph also shows the SIB (Severe Impairment Battery) change from the baseline, which is measured on a scale of 0-100. However, without knowing the context and purpose of the graph, it is difficult to provide a complete and accurate description.*
figure 9 - eb0ec3ab 9077 4d91 a965 4b8ed556951f 10

This appears to be a graph or chart related to a clinical trial, showing some sort of measurement or assessment of clinical improvement or decline over the course of 3-4 months of drug treatment. There are two groups being compared: one receiving Donepezil hydrochloride 10 mg/day and the other receiving a placebo. However, without proper context, it is not possible to determine the exact nature or significance of the data being presented.*
figure 10 - eb0ec3ab 9077 4d91 a965 4b8ed556951f 11
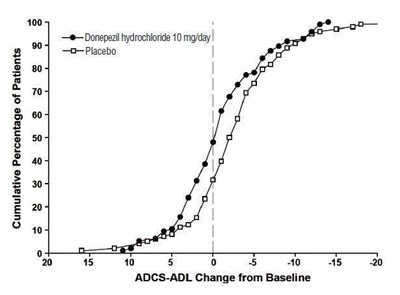
This chart displays the cumulative percentage of patients taking Donepezil hydrochloride 10 mg/day versus Placebo, in relation to the ADCS-ADL change from baseline. The graph shows that the Donepezil group outperformed the Placebo group in terms of improvement in ADL scores, with 100% of patients experiencing some degree of improvement compared to only around 50% in the Placebo group.*
figure 13 - eb0ec3ab 9077 4d91 a965 4b8ed556951f 14
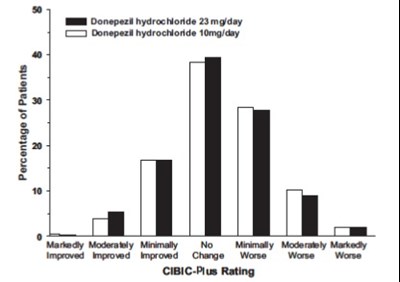
This is a table presenting the percentage of patients with varying degrees of improvement based on their ratings in the CIBIC-Plus system while taking different dosages of Donepezil hydrochloride (3mg/day or 10mg/day). There are no descriptions of what CIBIC-Plus rating system or the drug Donepezil is, so the context is unclear.*
5mg 90s - eb0ec3ab 9077 4d91 a965 4b8ed556951f 16

Each coated tablet of Donepezil Hydrochloride USP 5 g contains an ALM coating. The usual dosage is provided in accompanying information. The medicine should be stored at room temperature and in a container defined as D3P. The container should be kept safe from light, moisture, and high temperature. The product does not require refrigeration. The tablets are manufactured by SIBCO Pharmaceuticals, located in Tamilnadu, India. Pharmacist must dispense a patient information sheet for each patient. The text includes an NDC code for the medicine.*
5mg 500s - eb0ec3ab 9077 4d91 a965 4b8ed556951f 17

Product Name: Donepezil Hydrochloride Tablets, USP. Manufacturer: Strides Pharma Inc. NDC: 64380-907-07. Prescription only medicine. Batch/Lot No: THODO02225. Information sheet is enclosed in the package. Manufacturer's address: SIDCO Pharmaceutical Complex, Pharmacist at Kanchipuram, Tamil Nadu, India.*
10mg 90s - eb0ec3ab 9077 4d91 a965 4b8ed556951f 19

Each coated tablet contains 10mg of the USP Donepezil Hydrochloride. The usual dosage information is not available. The container is designed for light and moisture resistance and has specific storage instructions. Manufactured by SIBCO Pharmaceuticals at an address in Taniiads. Distributed with a specific NDC number as a prescription-only medicine. Instructions to dispense patient information sheet. The text contains errors and some information is unclear, indicating that this may not be a reliable source of information.*
10mg 500s - eb0ec3ab 9077 4d91 a965 4b8ed556951f 20

Each coated tablet of Donepez Hydrochloride USP contains 10mg. The usual dosage is mentioned in the accompanying information. The container is resistant and defined by U3P for dispensing at room temperature between 20 to 25° Celsius. The product is manufactured by Jimesa Sciences Pvt. Ltd. and distributed by Strides Pharma Inc. The provided NDG number is 64380-908-07. The text also includes an information sheet for the patient.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.