Product Images Losartan Potassium 100 Mg
View Photos of Packaging, Labels & Appearance
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 01
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 02
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 03
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 04
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 05
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 06
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 07
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 08
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 09
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 10
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 11
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 12
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 13
- 03375ec9 0e3a 40c2 8c77 de5d9fd21876 14
Product Label Images
The following 14 images provide visual information about the product associated with Losartan Potassium 100 Mg NDC 64380-935 by Strides Pharma Science Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
03375ec9 0e3a 40c2 8c77 de5d9fd21876 03

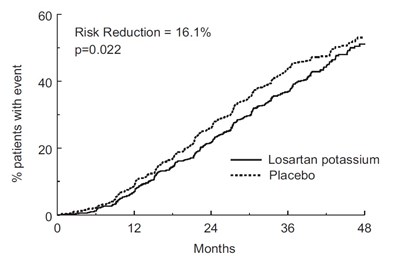
This is a chart showing the percentage of patients with fatal and non-fatal stroke in a study. However, there is not enough information to interpret the chart accurately.*
03375ec9 0e3a 40c2 8c77 de5d9fd21876 04

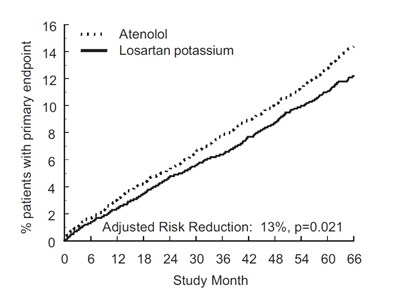
This appears to be a chart or table reporting the results of a study on the use of Losartan Potassium and Atenolol in patients with a primary composite stroke, comparing the rates of fatal vs non-fatal events. The data is stratified by age, gender, race, and presence of other health conditions. The table also includes hazard ratios and confidence intervals.*
03375ec9 0e3a 40c2 8c77 de5d9fd21876 07

Losartan Potassium Tablets, USP 25g is manufactured by Vined Lt Sciences Pvt. Ltd. The usual dosage is not available in this text, and it should be dispensed with caution. The product should be stored at temperatures not exceeding 30°C and 50 "t B6°). The text contains a lot of typographical errors and scattered information, making it challenging to comprehend its content.*
03375ec9 0e3a 40c2 8c77 de5d9fd21876 08

Each tablet contains Losartan Potassium USP. It is available in a pack size of 1000 film-coated tablets. The usual dosage is defined in the USP. The tablet should be protected from light and kept in a closed container. The manufacturer is Viimed Lite Sciences Private Limited. The pharmaceutical information leaflet is attached to the package, and it should be dispensed by the pharmacist. Distributed by Strides Pharma Inc. in NJ. This description is for Losartan Potassium tablets.*
03375ec9 0e3a 40c2 8c77 de5d9fd21876 09

This appears to be a label or packaging information for a drug called Losartan Potassium. The drug comes in tablets form and is classified under USP. It is unclear what dosage or strength it is as the information is not provided. The rest of the text is not readable and appears to contain errors.*
03375ec9 0e3a 40c2 8c77 de5d9fd21876 11

Each tablet of Losartan Potassium contains 50mg of USP. It is an Rx only drug that should be dispensed in a light-resistant container. The package insert should be consulted to determine the usual dosage. Store the drug at 25°C (77°F) with excursions allowed from 15° to 30°C (50° to 86°F) per the USP Controlled Release Tablets. It is produced and distributed by Vivimed Life Sciences Private Limited. Pharmacists are advised to dispense the drug with the patient information leaflet attached. The text also contains a lot number and a manufacturing date.*
03375ec9 0e3a 40c2 8c77 de5d9fd21876 14

This is a description of a medication called Losartan Potassium, which comes in tablet form with a 100mg dosage. The usual dosage information can be found in the package insert. It should be stored in a tightly sealed, light-resistant container. The medication can be stored at 25°C (77°F), with permitted temperature excursions of 15° to 30°C (50° to 86°F), according to the USP Controlled Room Temperature guidelines. The medication's ML number is TNOD002326, and it is produced by Vivimed Life Sciences, located in Tamilnadu, India. The medication should be kept away from light, and the NDC number is 64380-935-08. The medication package includes an attached patient information leaflet, and the manufacturer is Strides Pharma Inc.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.