Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
- Structure - donephcltabs figure 01
- Figure 1 - donephcltabs figure 02
- Figure 2 - donephcltabs figure 03
- Figrure 3 - donephcltabs figure 04
- Figure 4 - donephcltabs figure 05
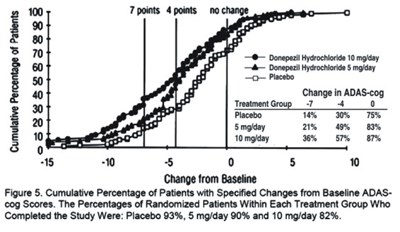
- Figure 5 - donephcltabs figure 06
- Figure 6 - donephcltabs figure 07
- Figure 7 - donephcltabs figure 08
- Figure 8 - donephcltabs figure 09
- Figure 9 - donephcltabs figure 10
- Figure 10 - donephcltabs figure 11
- Container Label - donephcltabs figure 12
- Container Label - donephcltabs figure 13
Product Label Images
The following 13 images provide visual information about the product associated with Donepezil Hydrochloride NDC 64679-312 by Wockhardt Usa Llc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - donephcltabs figure 02
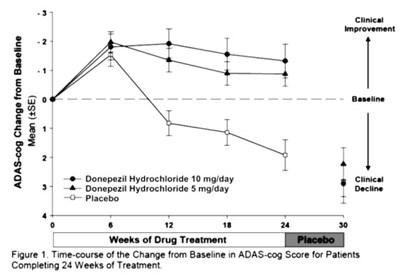
This is a graph displaying the time-course change from baseline in ADAS-cog score for patients completing 24 weeks of treatment with Donepezil Hydrochloride 10mg/day or Donepezil Hydrochloride 5mg/day or a Placebo. Clinical improvement is observed for the 10mg/day dosage, while a decline is shown for the 5mg/day dosage and placebo.*
Figure 2 - donephcltabs figure 03

The text provides a chart displaying the change in ADAS-cog in a treatment group taking Donepezil Hydrochloride 10mg/day, a treatment group taking Donepezil Hydrochioride 5mg/day, and a group taking placebo. Unfortunately, no description or context is available to further interpret the chart.*
Figrure 3 - donephcltabs figure 04

This text represents the results of a 24 week study evaluating the efficacy of Donepezil Hydrochloride at 5mg/day and 10mg/day in improving the CIBIC-plus rating score. The figure shows the frequency distribution of CIBIC plus scores at week 24.*
Figure 4 - donephcltabs figure 05
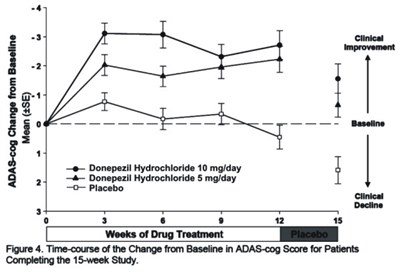
The text describes a clinical study evaluating the effectiveness of Donepezil Hydrochloride at 5mg/day and 10mg/day dosages compared to a placebo. The figure shown in the text depicts the time-course of change from baseline in ADAS-cog score after 15 weeks of drug treatment for patients who completed the study.*
Figure 6 - donephcltabs figure 07
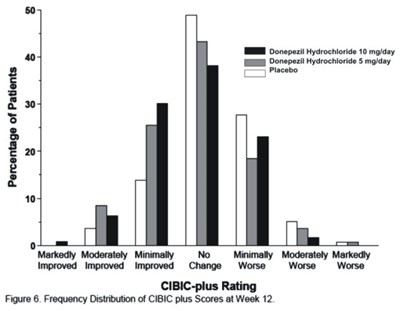
The text describes the dosages of Donepezil Hydrochloride, a medication used for treating dementia, in two different groups. There is also a chart showing the frequency distribution of CIBIC-plus ratings after twelve weeks of treatment. The chart is displaying scores of improvement, no change or worsening of symptoms.*
Figure 7 - donephcltabs figure 08

This is a graph showing the change from baseline in SIB score for patients completing 6 months of treatment with Donepezil Hydrochloride 10mg/day and placebo. The SIB score relates to clinical decline and ranges from -8 to 4.*
Figure 8 - donephcltabs figure 09
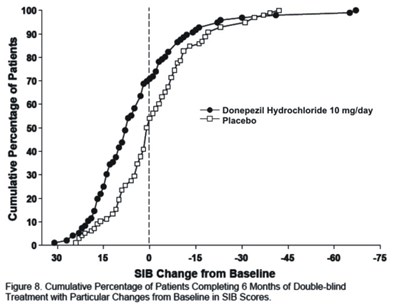
This is a graph showing the cumulative percentage of patients completing 6 months of double-blind treatment with either a 10mg/day dose of Donepezil Hydrochloride or Placebo. The graph shows the changes from baseline in SIB scores over time. The data points on the graph are not legible, so no further information can be provided.*
Figure 9 - donephcltabs figure 10
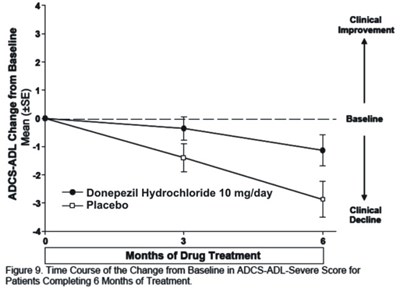
The text is a figure showing a time course of change from baseline in ADCS-ADL-Severe Score for patients completing 6 months of treatment with Donepezil Hydrochloride 10mg/day or Placebo Clinics, indicating a decline in both treatments.*
Figure 10 - donephcltabs figure 11

This appears to be medication instructions or dosage schedule. The medication is Donepezil Hydrochloride and the suggested dose is 10 mg per day. The remaining text seems to be a graph or chart showing the change in ADCS-ADL scores in patients completing 6 months of double-blind treatment.*
Container Label - donephcltabs figure 13

This is the prescribing information for Donepezil HCI 10 mg which is available in tablet form. The tablets contain the active ingredient donepezil hydrochloride. The recommended dosage and usage information is available in the accompanying prescribing information. The tablets should be dispensed in tight containers and stored at a temperature between 20°-25°C (68°-77°F). This product is manufactured and distributed by WOCKHARDT and comes in 30 tablet packs with NDC 64679-312-01.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.