Product Images Lisdexamfetamine Dimesylate
View Photos of Packaging, Labels & Appearance
- Figure 4 - Figure 4
- Figure 5 - Figure 5
- Figure 6 - Figure 6
- Figure 7 - Figure 7
- Figure 8 - Figure 8
- Lisdex Caps 10mg - Lisdex Caps 10mg bottle label
- Lisdex Caps 20mg - Lisdex Caps 20mg bottle label
- Lisdex Caps 30mg - Lisdex Caps 30mg bottle label
- Lisdex Caps 40mg - Lisdex Caps 40mg bottle label
- Lisdex Caps 50mg - Lisdex Caps 50mg bottle label
- Lisdex Caps 60mg - Lisdex Caps 60mg bottle label
- Lisdex Caps 70mg - Lisdex Caps 70mg bottle label
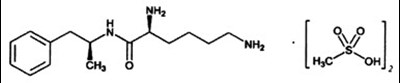
- Chemical Structure - vyvanse 01
- Figure 1 - vyvanse 02
- Figure 2 - vyvanse 03
- Figure 3 - vyvanse 04
Product Label Images
The following 16 images provide visual information about the product associated with Lisdexamfetamine Dimesylate NDC 64850-555 by Elite Laboratories, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 4 - Figure 4
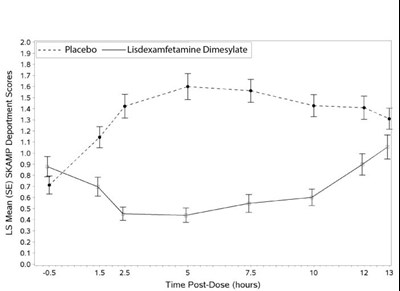
This text appears to show data related to SKAMP Deportment scores with LS Mean (SE) values for placebo and lisdexamfetamine Dimesylate at different time points post-dose. The values provided seem to represent scores at various time intervals.*
Figure 6 - Figure 6

The text indicates LS Mean (SE) PERMP Total Scores measured at different time points (0, 2, 4, and 8 hours post-dose) for both the Placebo and Lisdexamfetamine Dimesylate groups. The data suggests a comparison of the effects of the two treatments on PERMP Total Scores over time.*
Lisdex Caps 10mg - Lisdex Caps 10mg bottle label

This text seems to be a medication label for capsules containing Lisdexamfetamine. Each capsule contains Lisdexamfetamine dimesylate 10 mg. The manufacturer is Elite Laboratories, Inc. The capsules should be stored at 20° to 25°C (68° to 77°F) with excursions permitted to 15° to 30°C (59° to 86°F). The directions advise keeping the medication in a light-resistant container with a child-resistant closure. Each bottle contains 100 capsules. Pharmacist is advised to dispense an accompanying Medication Guide with each patient.*
Lisdex Caps 20mg - Lisdex Caps 20mg bottle label

The provided text seems to be a package insert for a medication called Lisdexamfetamine. Each capsule contains Lisdexamfetamine dimesylate 20 mg, manufactured by Elite Laboratories, Inc. It is recommended to store the medication at a temperature between 20° to 25°C (68° to 77°F). The expiration date of the medication is stated as 04/24. Pharmacists are advised to dispense the medication with a medication guide to each patient and ensure it is in child-resistant packaging. The medication is contained in 100 capsules per bottle.*
Lisdex Caps 30mg - Lisdex Caps 30mg bottle label

This text provides important information about a medication called Lisdexamfetamine, specifically the dosage, manufacturer (Elite Laboratories, Inc.), and storage instructions. The medication comes in capsule form, with each capsule containing 30 mg of Lisdexamfetamine dimesylate. The storage temperature ranges from 20°C to 25°C (68°F to 77°F) with excursions permitted up to 15° to 30°C (59°F to 86°F). It also mentions the need for a child-resistant closure and includes a medication guide for patients.*
Lisdex Caps 40mg - Lisdex Caps 40mg bottle label

This text appears to be information regarding the dosage instructions and storage of a medication called Lisdexamfetamine. The text includes details about the dosage, manufacturer (Elite Laboratories, Inc.), composition, storage conditions (20°C to 25°C), and packaging (100 capsules with a child-resistant closure). The medication guide needs to be dispensed to each patient along with the medication. The information also includes the National Drug Code (NDC) for this particular medication (64850-553-01).*
Lisdex Caps 50mg - Lisdex Caps 50mg bottle label

This text provides information about a medication called Lisdexamfetamine, in the form of capsules containing 50 mg of Lisdexamfetamine dimesylate. The manufacturer is Elite Laboratories, Inc. It is recommended to store the medication at a temperature between 20° to 25°C (68° to 77°F), with excursions permitted to 15° to 30°C (59° to 86°F). The medication should be dispensed in a tight, light-resistant container with a child-resistant closure. The package insert includes a medication guide for each patient.*
Lisdex Caps 60mg - Lisdex Caps 60mg bottle label

This text provides information about a medication containing Lisdexamfetamine dimesylate in capsules, each capsule containing 60 mg of the active ingredient. It also includes instructions for storage and dispensing by a pharmacist. The medication is manufactured by Elite Laboratories, Inc. and should be stored at a temperature between 20° to 25°C (68° to 77°F). Additionally, it mentions the NDC number for identification.*
Lisdex Caps 70mg - Lisdex Caps 70mg bottle label

This text appears to be a section from a medication package insert. It provides information on a capsule containing Lisdexamfetamine dimesylate 70 mg, manufactured by Elite Laboratories, Inc. The storage conditions are specified as 20° to 25°C (68° to 77°F) with excursions permitted to 15° to 30°C (59° to 86°F). The medication should be kept in a tight, light-resistant container with a child-resistant closure. The text includes details on the dosage and expiry date, as well as instructions for pharmacists to dispense the accompanying Medication Guide to each patient. This information is important for safe administration and storage of the medication.*
Figure 2 - vyvanse 03
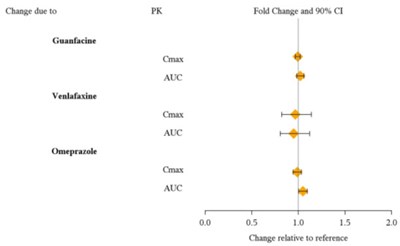
This text provides information on the change due to PK Fold Change and 90% Confidence Interval for the drugs Guanfacine, Venlafaxine, and Omeprazole. It includes details such as Cmax and Auc for each drug, with a graph showing changes relative to a reference point.*
Figure 3 - vyvanse 04

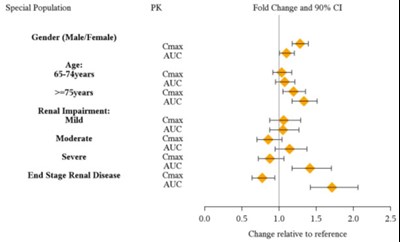
This description provides information on the PK (pharmacokinetic) fold change and 90% confidence interval for the drug Guanfacine when interacting with various drug substrates classified under different cytochrome P450 (CYP) enzymes such as CYP1A2, CYP2D6, CYP2C19, and CYP3A. The data includes parameters like Cmax (maximum concentration) and AUC (area under the curve) for these substrates. The chart visually represents the fold change relative to the reference drug.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.