Product Images Esomeprazole Magnesium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Esomeprazole Magnesium NDC 64980-632 by Rising Pharma Holdings, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
20mg-30s.jpg - 20mg 30s

This text provides information about Esomeprazole Magnesium Delayed-Release Capsules, USP. Each capsule contains 20mg of Esomeprazole. It includes advice for pharmacists to dispense a Medication Guide to each patient and mentions the usual adult dosage. The storage instructions recommend keeping the capsules between 15°C to 30°C. The manufacturer is CSPC Ouyi Pharmaceutical Co. in China, and the distributor is Rising Pharma Holdings in NJ, USA.*
40mg-30s - 40mg 30s

This is information about Esomeprazole 40mg delayed-release capsules manufactured by CSPC Ouyi Pharmaceutical Co., Ltd in Shijazhuang, Hebei, China. The capsules should be stored at 25°C (77°F) with excursions between 15° to 30°C (59° to 86°F). They are distributed by Rising Pharma Holdings, Inc. in Brunswick, NJ, with a suggested adult dosage provided in the package. It is advisable to keep the container tightly closed and out of reach of children. Pharmacists are instructed to dispense a medication guide along with the 30 capsules.*
Table-3 - Table 3

The text provides important information about potential drug interactions involving antiretrovirals, esomeprazole magnesium, and various other medications such as warfarin, methotrexate, clopidogrel, citalopram, cilostazol, digoxin, clarithromycin, amoxicillin, tacrolimus, and drugs dependent on gastric pH for absorption. It highlights the need to monitor and adjust dosages to prevent reduced efficacy, increased toxicity, or other adverse effects when these drugs are used concomitantly. Additionally, it mentions the importance of considering alternative medications in some cases.*
Table-4 - Table 4
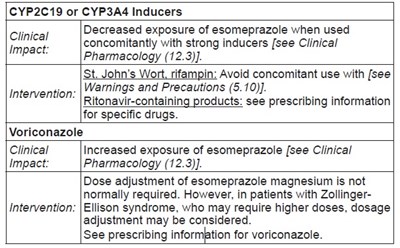
This information provides guidelines on interactions with CYP2C19 or CYP3A4 inducers such as St. John’s Wort, rifampin, and ritonavir-containing products when using esomeprazole. It highlights the potential decreased exposure of esomeprazole when used concomitantly with strong inducers and suggests avoiding the concurrent use of certain drugs. It also mentions Voriconazole and how it can increase exposure to esomeprazole, along with considerations for dose adjustment, especially in patients with specific conditions like Zollinger-Ellison syndrome. It is advised to refer to the prescribing information for specific details on voriconazole.*
esomeprazole-fig2 - esomeprazole fig2

This text appears to contain information related to a study involving Esomeprazole Magnesium at different doses (40 mg, 20 mg, 10 mg) and a placebo group. The number of participants (N) in each group is also provided. The data seems to be measured over multiple months, but specific details about the results or outcomes are not available.*
esomeprazole-fig3 - esomeprazole fig3

This is a description of a study showing the percentage of patients who maintained their condition while taking different doses of Esomeprazole Magnesium (40mg, 20mg, and 10mg) compared to a placebo. The study included 82 patients for both 40mg and 20mg doses and 77 patients for the 10mg dose and the placebo group.*
esomeprazole-fig5 - esomeprazole fig5

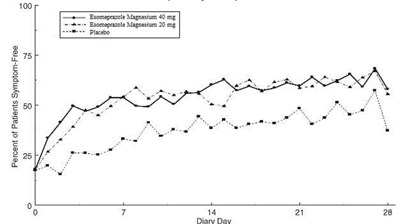
This text provides data on the percentage of patients who were symptom-free over a specific time period. It includes numbers corresponding to different medications or dosage strengths. The information seems to be related to recording symptoms on a diary over a period of several days.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.