Product Images Ipratropium Bromide And Albuterol Sulfate
View Photos of Packaging, Labels & Appearance
- b5387e98-figure-01 - b5387e98 figure 01
- b5387e98-figure-02 - b5387e98 figure 02
- b5387e98-figure-03 - b5387e98 figure 03
- b5387e98-figure-04 - b5387e98 figure 04
- carton-30ct-label - carton 30ct label
- carton-60ct-label - carton 60ct label
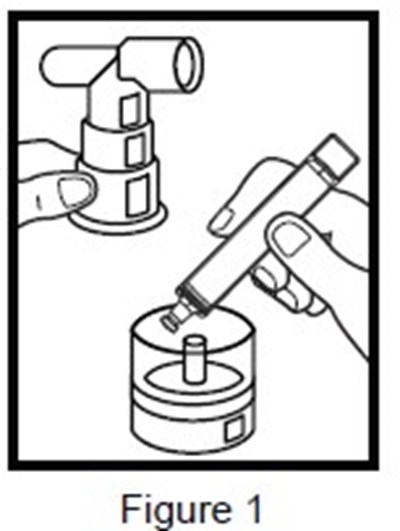
- figure-1 - figure 1
- figure-2 - figure 2
- figure-3 - figure 3
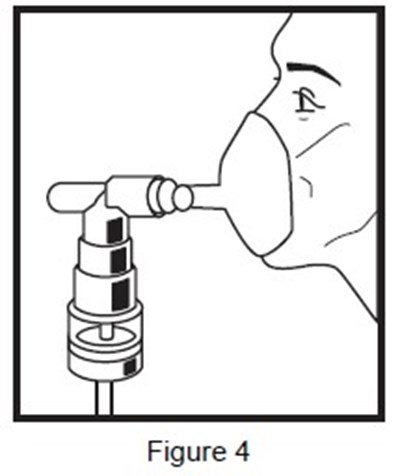
- figure-4 - figure 4
- vial label - vial label
Product Label Images
The following 11 images provide visual information about the product associated with Ipratropium Bromide And Albuterol Sulfate NDC 64980-645 by Rising Pharma Holdings, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
b5387e98-figure-04 - b5387e98 figure 04
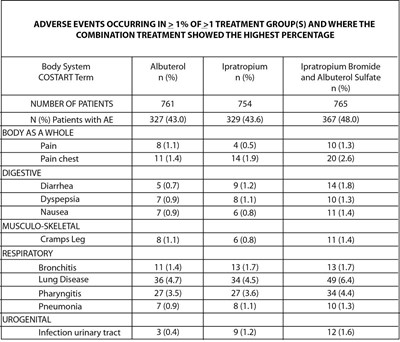
This data table shows the adverse events occurring in greater than 1% of more than one treatment group and illustrates the combination treatment group with the highest percentage for each event. The body system categories include Body as a Whole, Digestive, Musculo-skeletal, Respiratory, and Urogenital, with specific conditions and percentages of patients affected by each adverse event such as Pain, Diarrhea, Cramps Leg, Bronchitis, Lung Disease, and Infection urinary tract. This information can be helpful for healthcare professionals to evaluate and monitor potential side effects of treatment options.*
carton-30ct-label - carton 30ct label
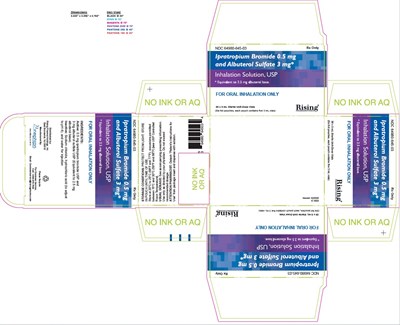
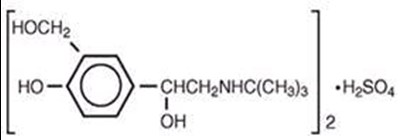
This text appears to be a description of a medication containing Ipratropium Bromide 0.5 mg and Albuterol Sulfate 3 mg in an inhalation solution form. The medication is for oral inhalation only and is used to treat conditions such as asthma or chronic obstructive pulmonary disease (COPD). It provides details on the active ingredients and dosage.*
carton-60ct-label - carton 60ct label
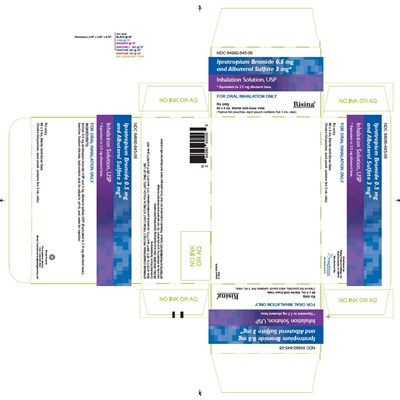
This is a prescription medication with the National Drug Code (NDC) 64980-245.00. It contains Ipratropium Bromide 0.5 mg and Albuterol Sulfate 3 mg for oral inhalation only. The medication is intended for respiratory use.*
vial label - vial label

This is a detailed label of a medication containing Ipratropium Bromide 0.5 mg and Albuterol Sulfate 3 mg per 3 mL vial. The medication is for oral inhalation only and should be used under the direction of a physician. It is stored between 20°C to 25°C and should be protected from light. Each vial should be used within one week after removing it from the foil pouch. The inactive ingredients include Sodium chloride, hydrochloric acid, and water for injection. The product is distributed by Rising Pharma Holdings, Inc. with customer service contact information provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.