Product Images Glycopyrrolate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Glycopyrrolate NDC 65145-104 by Caplin Steriles Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image2 - glycopyrrolate 02

Glycopyrrolate Injection, USP is a medication available in a 1 mL single-dose vial containing 0.2 mg/mL for intramuscular or intravenous use. It also contains benzyl alcohol. The product is manufactured by Caplin Steriles Limited in India, and its NDC number is 65145-102-01.*
image3 - glycopyrrolate 03

This is a medication called Glycopyrrolate Injection, which is available only with a prescription. It comes in 251 mL single-dose vials and can be administered intravenously or intramuscularly. The vial contains benzyl alcohol. The text mentions a "Bottom Lock," which may refer to a locking mechanism on the vial.*
image4 - glycopyrrolate 04
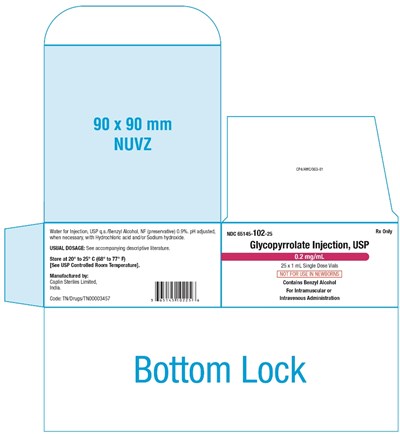
This is a description of a pharmaceutical product. The product is a solution available in individual vials of 90 x 90mm size for injection use only. It contains water for injection and 0.9% Benzyl Alcohol NF used as a preservative. Hydrochloric acid and/or Sodium hydroxide is used to adjust the pH. It is required to store the product at a controlled room temperature of 20° to 25° C (68° to 77° F). The usual dosage of the product is not mentioned and it is suggested to refer to the descriptive literature provided with the product. The product is manufactured by Gaplin Storls Ltd. India, and labeled with the code: TN/Drugs/TNOD0O3457 and the product name is Glycopyrrolate Injection, USP. The product is available in packs of 25x1ml single-dose vials for intramuscular or intravenous administration only, and it is advised for use by healthcare professionals only.*
image5 - glycopyrrolate 05

This is a prescription medication called Glycopyrrolate in injection form, with a concentration of 0.2 mg/mL. It is packaged in a single-dose vial of 2 mL, with the NDC number 65145-103-01. The medication contains Benzyl Alcohol and is intended for intramuscular or intravenous use. It is manufactured by Caplin Steriles Limited in India, with a specified lot number of 16.5x8.5 mm and a code of TN/Drugs/TN00003457. The Bip number given is NUVZ.*
image6 - glycopyrrolate 06
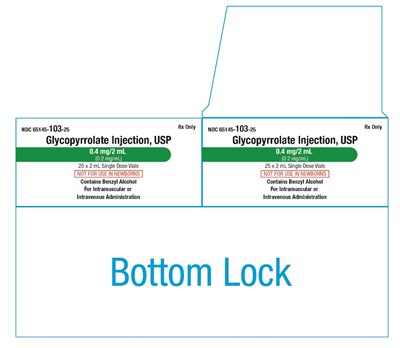
This is a description of a medication called Glycopyrrolate Injection, USP. It comes in a package of 25 single dose vials, each containing 0.4 mg/2 mL of the medication. The medication is intended for either intramuscular or intravenous administration and contains Benzyl Alcohol. The package is only available with a prescription.*
image8 - glycopyrrolate 08

This is a description of a product containing Water for Injection with Benzyl, recommended for prescription use only. It contains Alcohol as a preservative at 0.9%. The pH is adjusted using Glycopyrrolate where necessary. The manufacturer is Caplin Steriles Limited. The product also contains Hydrochloric acid and/or Sodium Injection and is not for use by newborns below 28 weeks of age. It is to be stored at 20°C to 25°C and used intramuscularly or intravenously.*
image9 - glycopyrrolate 09
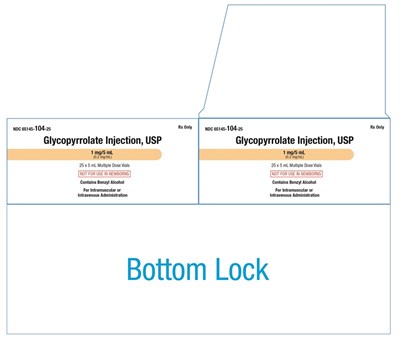
This is a description of the medication Glycopyrrolate Injection, USP. It is available in a 1 mg/5mL dose vial for intramuscular or intravenous administration and is only available by prescription. The medication contains benzyl alcohol and is not recommended for use in newborns. However, the rest of the text is not available or is illegible.*
image10 - glycopyrrolate 10

This is information about a medication called Glycopyrrolate Injection, USP 1 mg/5 mL. It should be stored at a controlled room temperature of 20-25°C and is available in a certain package size. The rest of the text is not legible due to errors in .*
image11 - glycopyrrolate 11

This is a description of the medical solution "Water for Injection, USP g.s./Benzyl Alcohol, NF (preservative) 0.9%". The solution's pH is adjusted with Hydrochloric acid and/or Sodium hydroxide when required. The regular dosage information is provided in accompanying descriptive literature. This solution is intended for "Intramuscular or Intravenous Use". The solution's storage instructions are to store it between 20°C and 25°C. The manufacturer of this solution is Caplin Steriles Limited. The lot number of this solution is "15x85mm". This solution should only be used by a prescription (Rx Only).*
image12 - glycopyrrolate 12

This is a description of a medication called Glycopyrrolate Injection, USP. It is a solution with a concentration of 4mg/20mL contained in 10 multiple dose vials. It is not safe for use in newborns and contains Benzyl Alcohol. It can be administered through intramuscular or intravenous means and requires a prescription.*
image13 - glycopyrrolate 13

This text is a medical description of an injection solution containing Water for Injection, USP, and Benzyl Alcohol, NF (preservative) 0.9%, which is used for intramuscular or intravenous administration. The dosage information is not provided in the text, and the accompanying descriptive literature should be referred to for this. The injection is not intended for use in newborns and contains Benzyl Alcohol. The injection is stored between 20° and 25° C (68° to 77° F). The manufacturer of this injection is Capin Steres Limited, located in India.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

