Product Images Lyllana
View Photos of Packaging, Labels & Appearance
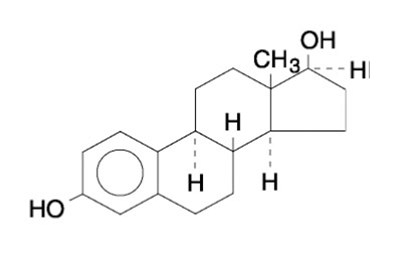
- formula - lyllana estradiol transdermal system 1
- Figure D - lyllana estradiol transdermal system 10
- Figure E - lyllana estradiol transdermal system 11
- Figure F - lyllana estradiol transdermal system 12
- Figure G - lyllana estradiol transdermal system 13
- 0.025 mg/day pouch - lyllana estradiol transdermal system 14
- 0.025 mg/day carton - lyllana estradiol transdermal system 15
- 0.0375 mg/day pouch - lyllana estradiol transdermal system 16
- 0.0375 mg/day carton - lyllana estradiol transdermal system 17
- 0.05 mg/day pouch - lyllana estradiol transdermal system 18
- 0.05 mg/day carton - lyllana estradiol transdermal system 19
- 3 layers - lyllana estradiol transdermal system 2
- 0.075 mg/day pouch - lyllana estradiol transdermal system 20
- 0.075 mg/day carton - lyllana estradiol transdermal system 21
- 0.1 mg/day pouch - lyllana estradiol transdermal system 22
- 0.1 mg/day carton - lyllana estradiol transdermal system 23
- Figure 1 - lyllana estradiol transdermal system 3
- Figure 2 - lyllana estradiol transdermal system 4
- Figure 3 - lyllana estradiol transdermal system 5
- Figure 4 - lyllana estradiol transdermal system 6
- figure a - lyllana estradiol transdermal system 7
- figure b - lyllana estradiol transdermal system 8
- Figure C - lyllana estradiol transdermal system 9
Product Label Images
The following 23 images provide visual information about the product associated with Lyllana NDC 65162-228 by Amneal Pharmaceuticals Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
0.025 mg/day pouch - lyllana estradiol transdermal system 14

LYLLANA is a transdermal system used for delivering estradiol in a dosage strength of 0.025 mg/day. The system contains 0.314 mg of estradiol and uses a combination of acrylic and silicone adhesive, oleyl alcohol, dipropylene glycol, povidone, polyester, and ethylene vinyl acetate copolymer film, and polyester release liner. It is important to note that the package is not child-resistant and should be kept out of reach of children. The system should be applied immediately after removing it from the pouch, and it should not be stored unpouched. The product is distributed by Amneal Pharmaceuticals LLC and must be stored between 20-25°C (68-77°F), with excursions allowed between 15-30°C (59-86°F).*
0.0375 mg/day pouch - lyllana estradiol transdermal system 16

LYLLA NA™ is a transdermal system that contains 0.470 mg of estradiol, USP that delivers 0.0375 mg/day. It is supplied in a 2.83 cm² system and contains inactive components such as acrylic adhesive, silicone adhesive, oleyl alcohol, dipropylene glycol, povidone, polyester and ethylene vinyl acetate copolymer film, polyester release liner. The package is not child-resistant and must be kept out of the reach of children. LYLLA NA™ must be applied immediately upon removal from the pouch and must not be stored unpouched. It is distributed by Amneal Pharmaceuticals LLC and must be stored at 20° to 25°C (68° to 77°F) with some excursions permitted.*
0.05 mg/day carton - lyllana estradiol transdermal system 19

This is a description of a medication product. The product consists of 8 systems, each containing 3.78 ounces which controls 0.627 grams of Estadio, LS. The product is for transdermal use only and is packaged with important instructions. Dosage and administration information is provided in the package insert. Information on inactive components is provided. The product is distributed by Aveal Pharmaceuticals LLC in NJ, USA. The rest of the text is unclear and not readable due to errors.*
3 layers - lyllana estradiol transdermal system 2

This is a product description for a type of adhesive that contains the hormone Estradiol. The product also features a protective liner and a backing material, although specific details about these components are not provided.*
0.075 mg/day pouch - lyllana estradiol transdermal system 20

7LYLLANA is a prescription-only estradiol transdermal system that delivers 0.075 mg/day. Each system contains 0.940 mg of estradiol, USP, along with inactive components like acrylic adhesive, silicone adhesive, oleyl alcohol, dipropylene glycol, povidone, polyester, and ethylene vinyl acetate copolymer film. The package is not child-resistant, and the system should be applied immediately upon removal from the pouch, as it should not be stored unpouched. It should be stored at a controlled room temperature of 20° to 25°C (68° to 77°F), with excursions permitted between 15° to 30°C (59° to 86°F). The product is distributed by Amneal Pharmaceuticals LLC.*
0.1 mg/day pouch - lyllana estradiol transdermal system 22

7LYLLA NA™ NDC 651 82~228~047 is a prescription-only transdermal system that delivers 0.1 mg/day of estradiol USP. Each system is 7.55 cm2 and contains 1.253 mg of estradiol USP along with inactive components such as acrylic adhesive, silicone adhesive, oleyl alcohol, dipropylene glycol, povidone, polyester, and ethylene vinyl acetate copolymer film, polyester release liner. It is important to keep this package out of the reach of children and apply it immediately upon removal from the pouch. Do not store unpouched. The system should be stored at 20° to 25°C (68° to 77°F) with excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. It is distributed by Amneal Pharmaceuticals LLC in Bridgewater, NJ.*
Figure 2 - lyllana estradiol transdermal system 4

The text describes data related to a reduction from a certain baseline, measured in an unknown unit called "an(sD)". There is also information about a specific dosage of "0.0375 mg/day" of something called "Vivete". The word "placebo" is mentioned, and it seems to indicate some sort of statistical difference between the effects of this placebo and the effects of Vivelle an. However, without more context or information, it's difficult to determine the nature of the study, what Vivete and Vivelle are, or what the reduction from baseline actually represents.*
figure b - lyllana estradiol transdermal system 8

LYLLANA is a pharmaceutical product containing the active ingredient estradiol. The given characters "ol ywom.5P)" are not readable and may be a result of incorrect .*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.