Product Images Stimufend
View Photos of Packaging, Labels & Appearance
- Fresenius Kabi Logo - sti04 0001 01
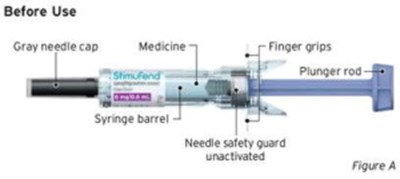
- Figure A - sti04 0001 02
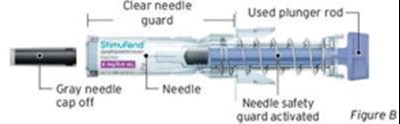
- Figure B - sti04 0001 03
- Figure C - sti04 0001 04
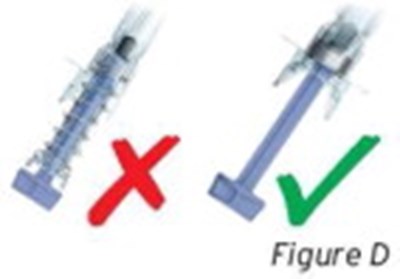
- Figure D - sti04 0001 05
- Figure E - sti04 0001 06
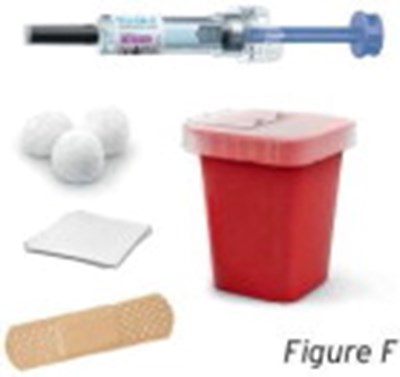
- Figure F - sti04 0001 07
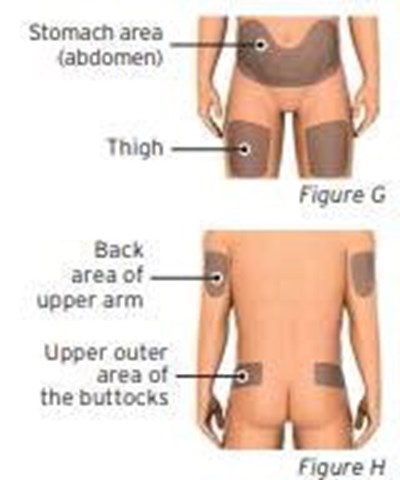
- Figures G and H - sti04 0001 08
- Figure I - sti04 0001 09
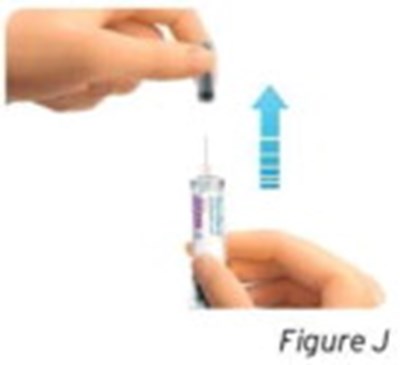
- Figure J - sti04 0001 10
- Figure K - sti04 0001 11
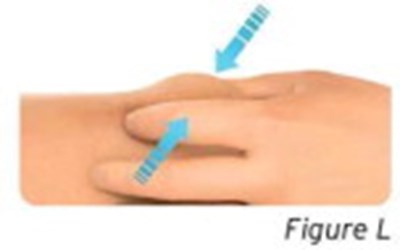
- Figure L - sti04 0001 12
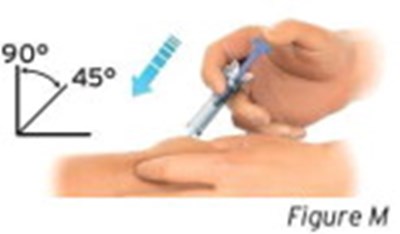
- Figure M - sti04 0001 13
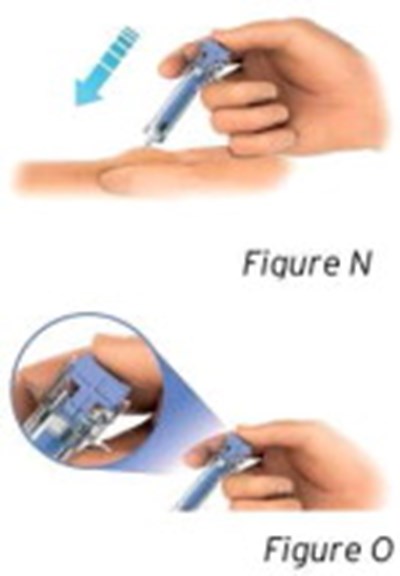
- Figures N and O - sti04 0001 14
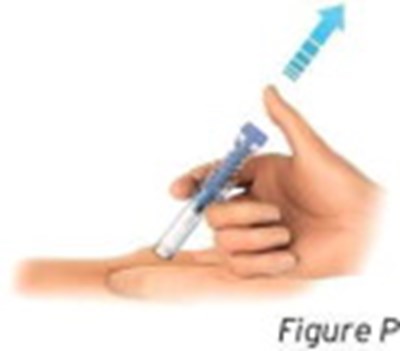
- Figure P - sti04 0001 15
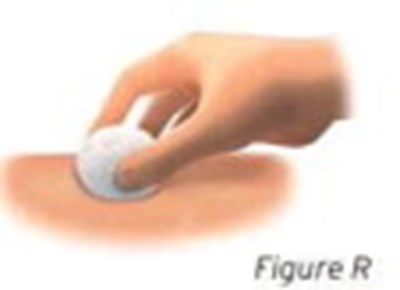
- Figure Q - sti04 0001 16
- Figure R - sti04 0001 17
- Fresenius Kabi Logo - sti04 0001 18
- PACKAGE LABEL – PRINCIPAL DISPLAY – STIMUFEND –One 0.6 mL Single-Dose Prefilled Syringe CARTON PANEL - sti04 0001 19
- PACKAGE LABEL – PRINCIPAL DISPLAY – STIMUFEND – 0.6 mL Single-Dose Prefilled Syringe BLISTER PACK LABEL - sti04 0001 20
- PACKAGE LABEL – PRINCIPAL DISPLAY – STIMUFEND – 0.6 mL Single-Dose Prefilled SYRINGE LABEL - sti04 0001 21
Product Label Images
The following 21 images provide visual information about the product associated with Stimufend NDC 65219-371 by Fresenius Kabi Usa, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE LABEL – PRINCIPAL DISPLAY – STIMUFEND –One 0.6 mL Single-Dose Prefilled Syringe CARTON PANEL - sti04 0001 19

PACKAGE LABEL – PRINCIPAL DISPLAY – STIMUFEND – 0.6 mL Single-Dose Prefilled Syringe BLISTER PACK LABEL - sti04 0001 20

Fresenius Kabi's StimuFend is a subcutaneous injection containing Pegylated Recombinant Methionyl Human Granulocyte Colony Stimulating Factor, used to treat low white blood cell count. It is stored refrigerated, protected from light, and is contained in a pre-filled syringe. This product is manufactured by Fresenius Kabi USA and has no U.S. standard potency. The given text contains necessary information about the product, precautions, and its manufacturing details.*
PACKAGE LABEL – PRINCIPAL DISPLAY – STIMUFEND – 0.6 mL Single-Dose Prefilled SYRINGE LABEL - sti04 0001 21
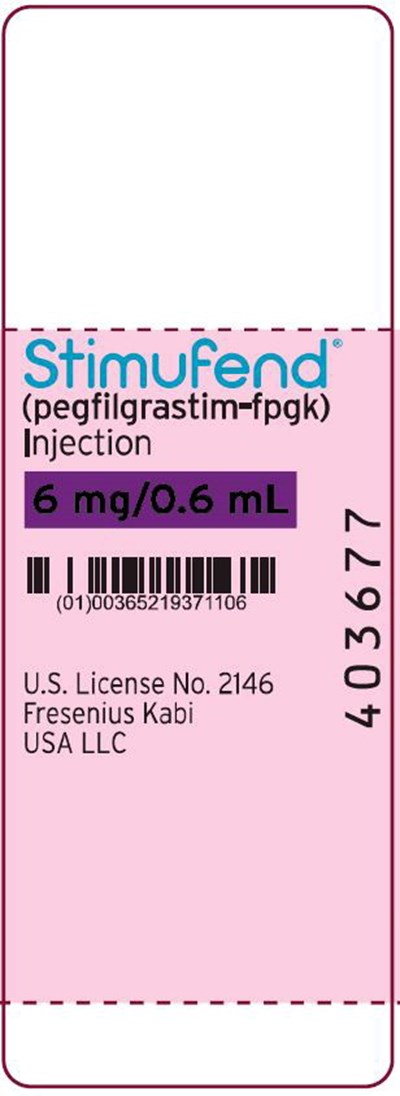
Stimufend' is an injection medication with the active ingredient pegfilgrastim=fpgk. It is U.S. licensed with the number 2146 and produced by Fresenius Kabi USA LLC. The dosage and usage instructions are not available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.