FDA Label for Diphenhydramine Hydrochloride
View Indications, Usage & Precautions
Diphenhydramine Hydrochloride Product Label
The following document was submitted to the FDA by the labeler of this product Himprit Pharmachem Pvt Ltd. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient (In Each Tablet)
Diphenhydramine HCl 25 mg
Purpose
Antihistamine
Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
- temporarily relieves these symptoms due to the common cold:
- runny nose
- sneezing
Directions
- take every 4 to 6 hours
- do not take more than 6 times in 24 hours
| adults and children 12 years and over | 1 to 2 tablets |
| children 6 to under 12 years | 1 tablet |
| children under 6 years | do not use this product in children under 6 years of age |
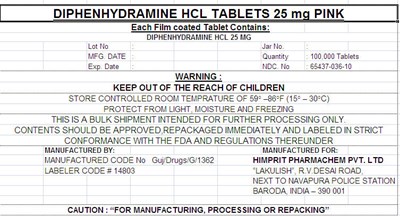
Principal Display Panel - 25 Mg Label
DIPHENHYDRAMINE HCL TABLETS 25 mg PINK
Each Film coated Tablet Contains:
DIPHENHYDRAMINE HCL 25 MG
| Lot No | : | Jar No. | : | ||
| MFG. DATE | : | Quantity | : 100,000 Tablets | ||
| Exp. Date | : | NDC. No | : 65437-036-10 |
WARNING :
KEEP OUT OF THE REACH OF CHILDREN
STORE CONTROLLED ROOM TEMPRATURE OF 59° –86°F (15° – 30°C)
PROTECT FROM LIGHT, MOISTURE AND FREEZING
THIS IS A BULK SHIPMENT INTENDED FOR FURTHER PROCESSING ONLY.
CONTENTS SHOULD BE APPROVED,REPACKAGED IMMEDIATELY AND LABELED IN STRICT
CONFORMANCE WITH THE FDA AND REGULATIONS THEREUNDER
MANUFACTURED BY:
MANUFACTURED CODE No Guj/Drugs/G/1362
LABELER CODE # 14803
MANUFACTURED FOR:
HIMPRIT PHARMACHEM PVT. LTD
"LAKULISH", R.V.DESAI ROAD,
NEXT TO NAVAPURA POLICE STATION
BARODA, INDIA – 390 001
CAUTION : "FOR MANUFACTURING, PROCESSING OR REPACKING"
* Please review the disclaimer below.