Product Images Relistor
View Photos of Packaging, Labels & Appearance
- figure1 - figure1
- figurea2.jpg - figurea2
- figurea3.jpg - figurea3
- figurebandc1.jpg - figurebandc1
- figurebandc2.jpg - figurebandc2
- figurebandc3.jpg - figurebandc3
- figured1.jpg - figured1
- figured2.jpg - figured2
- figured3.jpg - figured3
- figuree1.jpg - figuree1
- figuree2.jpg - figuree2
- figuree3.jpg - figuree3
- figuref1.jpg - figuref1
- figuref2.jpg - figuref2
- figuref3.jpg - figuref3
- figureg1.jpg - figureg1
- figureg2.jpg - figureg2
- figureg3.jpg - figureg3
- figureh1.jpg - figureh1
- figureh2.jpg - figureh2
- figureh3.jpg - figureh3
- figurei1.jpg - figurei1
- figurei2.jpg - figurei2
- figurei3.jpg - figurei3
- figurej2.jpg - figurej2
- figurej3.jpg - figurej3
- figurek1.jpg - figurek1
- figurek2.jpg - figurek2
- figurek3.jpg - figurek3
- figurel2.jpg - figurel2
- figurel3.jpg - figurel3
- figurem2.jpg - figurem2
- figurem3.jpg - figurem3
- figuren2.jpg - figuren2
- figuren3.jpg - figuren3
- figureo2.jpg - figureo2
- figureo3.jpg - figureo3
- figurep2.jpg - figurep2
- figurep3.jpg - figurep3
- figureq2.jpg - figureq2
- figureq3.jpg - figureq3
- figurer2.jpg - figurer2
- figurer3.jpg - figurer3
- figures2.jpg - figures2
- figures3.jpg - figures3
- figuret3.jpg - figuret3
- figuretandu2.jpg - figuretandu2
- figurev2.jpg - figurev2
- relistorcarton1 - image 01
- chemstructure - image 02
- figurej2 - image 03
- image 04
- image 05
- image 06
- image 07
- image 08
- image 09
- image 10
- image 11
- image 12
- image 13
- syringe1.jpg - syringe1
Product Label Images
The following 62 images provide visual information about the product associated with Relistor NDC 65649-150 by Salix Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure1 - figure1
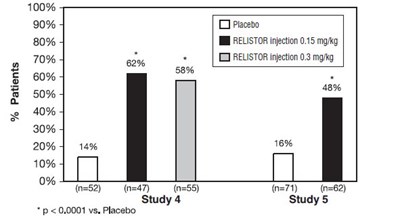
This appears to be a table with data on various treatments and their effectiveness for patients. Not enough context is given to determine what condition or illness is being treated.*
figurea2.jpg - figurea2
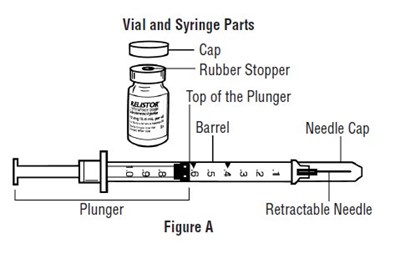
This is a diagram of different parts of a vial and syringe, including a cap, rubber stopper, plunger, needle, and barrel needle cap. The figure A shows a retractable needle plunger.*
figurea3.jpg - figurea3
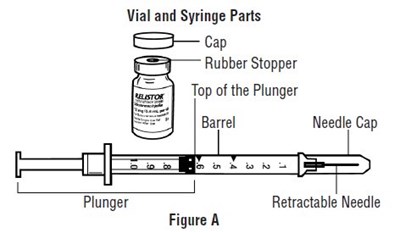
This appears to be a diagram or image of parts related to vials and syringes. The text indicates various components, including a rubber stopper, top of the plunger, barrel, needle cap, and retractable needle. The image shows a labeled diagram with these parts identified. No additional information or context is available.*
figurej2.jpg - figurej2
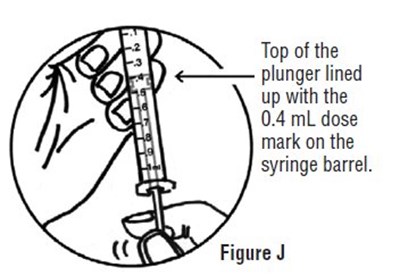
The text appears to be a technical instruction for administering medication through a syringe. It describes how to align the plunger with the 0.4 mL dose mark on the syringe barrel. The text is accompanied by a figure demonstrating the correct alignment.*
image 07

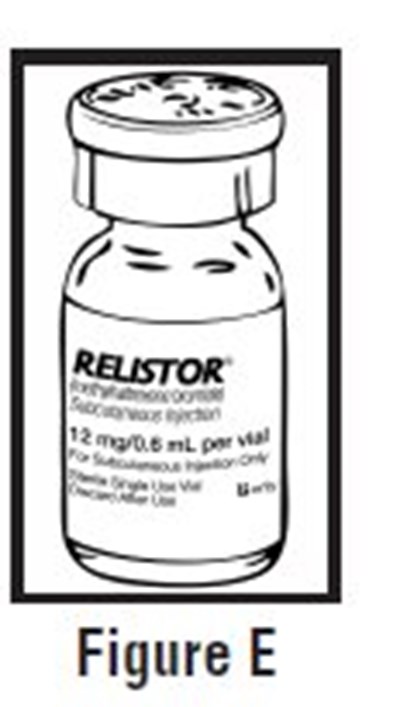
This is a description of a medication called RELISTOR containing pre-filled syringes of methylnaltrexone bromide for subcutaneous injection. The package includes seven syringes and four inserts with prescribing information, instructions, and a medication guide. Each syringe contains 12 mg of methylnaltrexone bromide, sodium chloride, edstate calcium disodium, and glycine hydrochloride. Dosage and administration may vary by patient weight. The medication should be kept at controlled room temperature and should not be frozen. The medication is manufactured by Progenics Pharmaceutical and is a trademark of Salix Pharmaceuticals. A pharmacist should dispense the medication guide to each patient.*
image 13
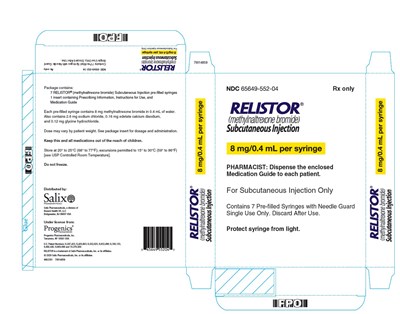
The text describes a package containing seven pre-filled syringes of RELISTOR® (methylnaltrexone bromide) Subcutaneous Injection for administration in patients. Each syringe contains 8 mg of methylnaltrexone bromide in 0.4 mL of water, and the dose may vary by patient weight. The package insert contains dosage and administration instructions, prescribing information, and a medication guide. The medication should be kept out of the reach of children and stored between 20°C to 25°C (68°F to 77°F) with excursions permitted to 15°C to 30°C (59°F to 86°F). The medication should not be frozen, and it is distributed by Salix.*
syringe1.jpg - syringe1
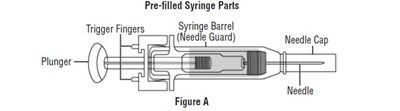
The text describes a pre-filled syringe with parts including a syringe barrel, a needle cap, a needle guard, a plunger, and a needle. The image labeled "Figure A" may illustrate these parts.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.