FDA Label for Mouth Sore Relief Applicator
View Indications, Usage & Precautions
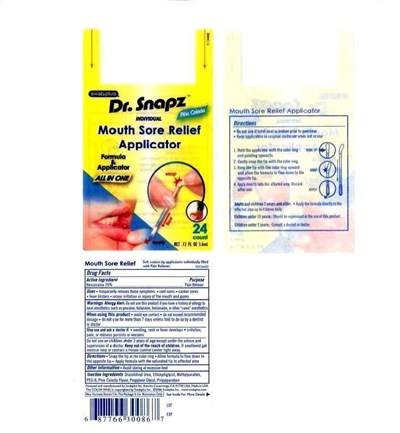
Mouth Sore Relief Applicator Product Label
The following document was submitted to the FDA by the labeler of this product Swabplus Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Drug Fact
Active Ingredient
Benzocaine 20%
Purpose
Pain Reliever
Uses
temporarily relieves these symptoms. cold sores. scanker sores. fever blisters. minor irritation or injury of the mouth and gums.
Warning Allergy Alert
Do not use this product if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine, or other "caine" anesthetics.
When Using This Product
avoid eye contact. do not exceed recommended dosage. do not use more than 7 days unless told to do so by a dentist or doctor.
Stop Use And Ask A Doctor If
swelling. rash or fever develops, irritation, pain, or redness persists or worsen.
Do not use on children under 2 years except underteh advice and supervision of a doctor.
Keep Out Of The Reach Of Children
if swallowed get medical help or contact a Poison Contro;mCenter right away.
Directions
Snap the tip at the color ring. Allow formula to flow down to the opposite tip. Apply formula with the saturated tip to affected area
Other Information
Avoid storaging at excessive heat.
Inactive Ingredient
Polyethylene glycol 400 (PEG-8), Glycerin, Saccharin sodium, water, pina Colada
Package Label.Principal Display Panel
Image of carton label
* Please review the disclaimer below.