Product Images Glyburide And Metformin Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Glyburide And Metformin Hydrochloride NDC 65841-825 by Zydus Lifesciences Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure - de7b5693 0366 4754 ad17 5e9d14563fb5 02
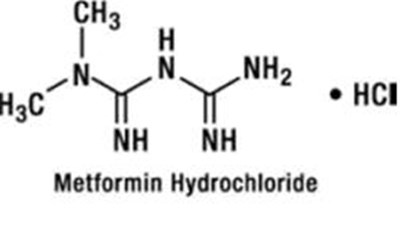
Metformin Hydrochloride is a medication that is abbreviated as "i ow", with additional characters that are not readable through . The medication is likely used to treat type 2 diabetes, but further information is not provided.*
figure - de7b5693 0366 4754 ad17 5e9d14563fb5 03

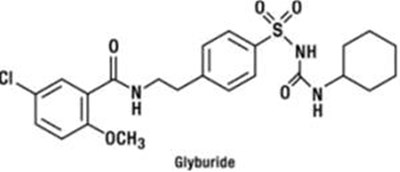
Glyburide and Metformin Hydrochloride Tablets are available in a pack of 100 tablets as U.S.P medication. Each 2ch i coated tablet contains 125mg of Giyouride, USP, and 250mg of Meformin Hyroctione, USP. The medication needs to be stored at a temperature of 20°C to 25°C. The tablets need to be dispensed to each patient separately, and the package insert needs to be provided along with it. The drug needs to be kept out of children's reach. Manufactured by Catia Healfcare L. in India.*
figure - de7b5693 0366 4754 ad17 5e9d14563fb5 04

This is a description for a medication called "Glyburide and Metformin Hydrochloride Tablets". The tablets contain 2.5 mg of glyburide and 500 mg of metformin hydrochloride and come in a container of 100 tablets. This medication should be dispensed by a pharmacist along with patient information. It is recommended that this medication be stored between 20°C to 25°C and be kept out of the reach of children. It is manufactured by Catia Healthcare Ltd. in Baddi, India. The usual dosage should be determined by consulting the package insert for complete prescribing information.*
figure - de7b5693 0366 4754 ad17 5e9d14563fb5 05

ZyGenerics Glyburide and Metformin Hydrochloride Tablets, USP 5 mg/500 mg. Each film-coated tablet contains Giyburide, USP: 5mg and Metiormin Hydrochioride, USP: 500mg. It is recommended to dispense patient information separately to each patient. Store in a tight container (USP) at 20° to 25°C (68" lo 77°F) [See: USP Controlled Room Temperature]. This drug should be kept out of the reach of children. Usual Dosage: See package insert for complete prescribing information. Manufactured by Cadla Healthcare Ltd, Baddi India.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.