Product Images Topiramate
View Photos of Packaging, Labels & Appearance
- Figure 1: Kaplan-Meier Estimates of Cumulative Rates for Time to First Seizure - topiramate fig1
- Table 12: Efficacy Results in Double-Blind, Placebo-Controlled, Add-On Epilepsy Trials - topiramate fig2
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 25 mg (60 Tablets Bottle) - topiramate fig3
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg (60 Tablets Bottle) - topiramate fig4
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg (60 Tablets Bottle) - topiramate fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg (60 Tablets Bottle) - topiramate fig6
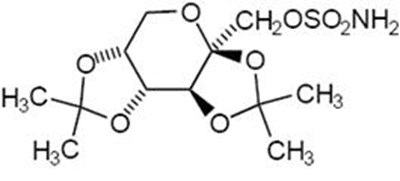
- chemical structure - topiramate str
Product Label Images
The following 7 images provide visual information about the product associated with Topiramate NDC 65862-171 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1: Kaplan-Meier Estimates of Cumulative Rates for Time to First Seizure - topiramate fig1

The text represents a graph showing cumulative rates for time to the first seizure for two groups, one taking topiramate 50 mg/day (N-234) and the other taking topiramate 400 mg/day (N-236), plotted against time in days up to 500.*
Table 12: Efficacy Results in Double-Blind, Placebo-Controlled, Add-On Epilepsy Trials - topiramate fig2

This appears to be a table with data from a study involving a medication called Topiramate. The table shows groups of participants who were given different doses of the medication or a placebo (shown as "placsbo"). The numbers in the parentheses indicate the total number of participants in each group. The asterisks (*) may be indicating statistically significant results, with the key at the bottom showing that "*p<0.01" and "**p<0.001" indicate different levels of significance. Without access to more context or clearer , it is difficult to provide a full description or interpretation of the data.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 25 mg (60 Tablets Bottle) - topiramate fig3

These are Topiramate Tablets USP with NDC code 65862-171-60. Each tablet contains 25 mg of Topiramate USP and is manufactured by Aurobindo Pharma USA, Inc. in India. The product information can be found in the accompanying literature, and the Pharmacist should dispense it along with the medication guide. The tablets should be stored in a tight container at room temperature and protected from moisture.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg (60 Tablets Bottle) - topiramate fig4

Topiramate is a medication used to treat seizures and prevent migraines. This specific product is manufactured by Aurobindo Pharma USA, Inc. and contains 60 tablets of 50mg each. The medication guide needs to be dispensed separately. The product should be stored in a tight container, protected from moisture, and kept at 20° to 25°C (68° to 77°F) with excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Recommended usage should be followed as per the accompanying literature.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg (60 Tablets Bottle) - topiramate fig5

This is a description of a medication with the NDC code 65862-173-60 labeled as Topiramate Tablets USP with a strength of 100 mg. It is distributed by Aurobindo Pharma USA, Inc. The medication guide is dispensed separately, and the usual dosage and other relevant prescribing information are included in the literature accompanying the product. It should be stored in a tightly closed container at 20-25°C, with excursions allowed between 15-30°C. The medicine must be safeguarded against moisture.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg (60 Tablets Bottle) - topiramate fig6

Topiramate Tablets USP with a strength of 200 mg and a quantity of 60 tablets per bottle. For dose and prescribing information, refer to the accompanying product literature. Distributed by Aurobindo Pharma USA, Inc. The medication guide should be provided separately. Store in a tightly closed container at room temperature and protect from moisture. The code for the product is TS/DRUGS/19/1993. This is for prescription use only.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.