Product Images Lamotrigine
View Photos of Packaging, Labels & Appearance
- Figure 1 - lamotrigine fig1
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg (100 Tablet Bottle) - lamotrigine fig10
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg Blister Carton (10 x 10 Unit-dose) - lamotrigine fig11
- Figure 2 - lamotrigine fig2
- Figure 3 - lamotrigine fig3
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 25 mg (100 Tablet Bottle) - lamotrigine fig4
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 25 mg Blister Carton (10 x 10 Unit-dose) - lamotrigine fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg (100 Tablet Bottle) - lamotrigine fig6
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg Blister Carton (10 x 10 Unit-dose) - lamotrigine fig7
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 150 mg (100 Tablet Bottle) - lamotrigine fig8
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 150 mg Blister Carton (10 x 10 Unit-dose) - lamotrigine fig9
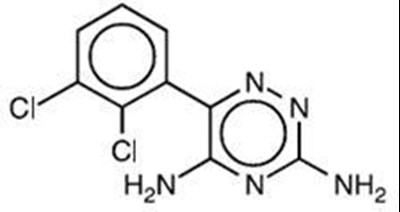
- Chemical Structure - lamotrigine str
Product Label Images
The following 12 images provide visual information about the product associated with Lamotrigine NDC 65862-229 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - lamotrigine fig1
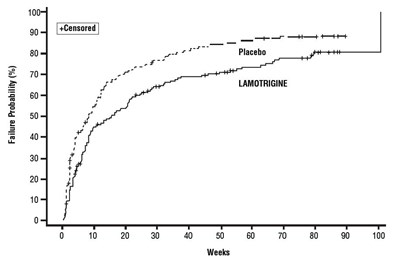
This data appears to be a graph displaying the failure probability percentage for a medication called Lamotrigine over the course of several weeks. The graph shows the percentage starting at 0 and gradually increasing up to 100, with a censored data point in between. Unfortunately, without more context or labeling on the graph, it is unclear what exactly constitutes "failure" or what application the medication is used for.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg Blister Carton (10 x 10 Unit-dose) - lamotrigine fig11
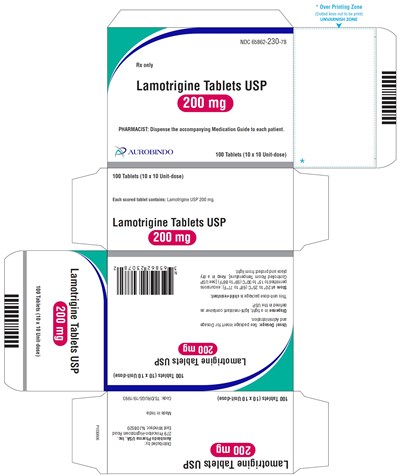
Lamotrigine Tablets USP are a prescription medicine containing 200 mg of lamotrigine used in adults with epilepsy and bipolar disorder. The medication guide should be given to each patient as instructed by the pharmacist. The text contains multiple numbers and codes related to the medication, packaging, and distribution which likely corresponds to a specific brand or product of Lamotrigine Tablets USP.*
Figure 2 - lamotrigine fig2

This appears to be a graph or chart displaying the failure probability percentage for a drug called lamotrigine over the course of several weeks. There is also a mention of "censored TR" which may refer to data that has been left out or condensed for analysis. However, without additional context or a clearer image, it is difficult to provide a more detailed description.*
Figure 3 - lamotrigine fig3
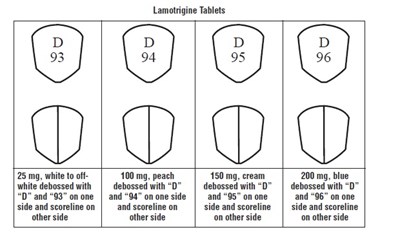
Lamotrigine Tablets are available in 25mg, 100mg, 150mg and 200mg doses. They are colored off-white, peach, cream and blue respectively and are imprinted with a debossed letter "D" on one side and a different number on the other side.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 25 mg (100 Tablet Bottle) - lamotrigine fig4

This is a description of Lamotrigine USP 25 mg tablets by Aurobindo Pharma USA. The tablets are scored and should be dispensed in a tight, light-resistant container as defined in the USP. The usual dosage should be determined by referring to the package insert. The tablets should be stored at a temperature range of 20°C to 25°C (68°F to77°F), and protected from light. The pharmacist should provide each patient with the accompanying medication guide. Unfortunately, further information is not available due to the nature of the text which appears to be a scanned document.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 25 mg Blister Carton (10 x 10 Unit-dose) - lamotrigine fig5
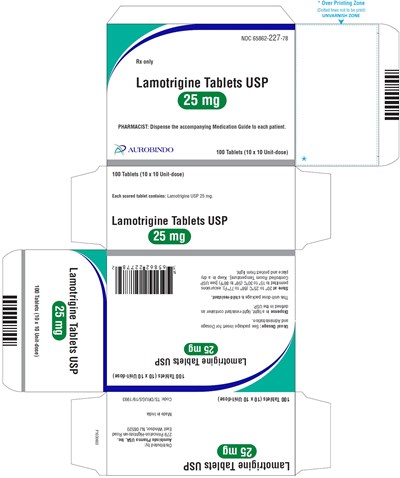
This is a medication label for Lamotrigine Tablets USP, a prescription medication used to treat seizures and bipolar disorder. The label lists the medication's name, dosage amount, and NDC number. Additionally, it instructs pharmacists to provide a medication guide to each patient. There is a warning not to print anything in the overprinting zone. The rest of the scanned text is not legible.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg (100 Tablet Bottle) - lamotrigine fig6

This is a label of Lamotrigine tablets, each containing 100 mg of USP. It is distributed by Aurobindo Pharma USA, Inc. The usual dosage instructions are present in the package insert. The tablets should be stored in a tight, light-resistant container and in a dry place, with a permitted temperature range of 20° to 25°C (68° to 77°F). The medication guide should be dispensed to each patient by the pharmacist, and the over printing zone coding area should not be printed.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg Blister Carton (10 x 10 Unit-dose) - lamotrigine fig7
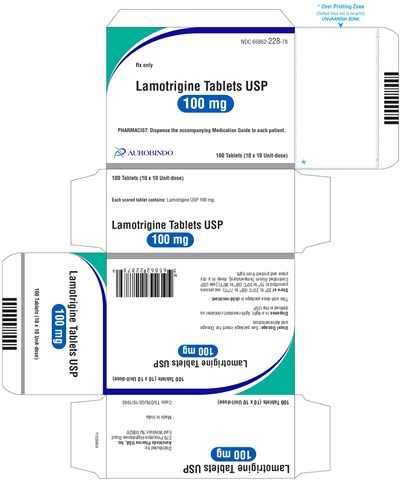
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 150 mg (100 Tablet Bottle) - lamotrigine fig8

Lamotrigine Tablets USP are available in a pack containing 100 tablets, with each scored tablet containing 150 mg of Lamotrigine USP. The usual dosage administration can be seen in the package insert. Pharmacists are advised to dispense the accompanying medication guide to patients and store this medication in a dry place and protect it from light. This drug is distributed by Aurobindo Pharma USA, Inc., made in India, and should be dispensed in a tight, light-resistant container as defined in the USP.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 150 mg Blister Carton (10 x 10 Unit-dose) - lamotrigine fig9
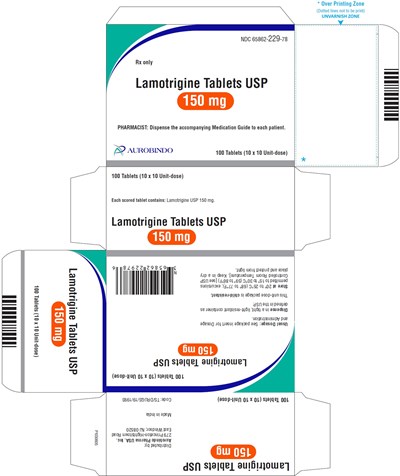
This appears to be a pharmaceutical label for the drug Lamotrigine, containing information on its dosage and administration. The label bears the name of the drug, the number of tablets and their composition, and indicates that it is for prescription-only use. The text's formatting suggests the presence of an overprinting and an unvarnished zone, and reminds pharmacists to dispense a medication guide to patients. A significant portion of the text, however, contains encoded strings that cannot be interpreted into meaningful language.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.