Product Images Rosuvastatin Calcium
View Photos of Packaging, Labels & Appearance
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg (30 Tablets Bottle) - rosuvastatin fig1
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg Blister Carton (10 x 6 Unit-dose) - rosuvastatin fig2
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 10 mg (30 Tablets Bottle) - rosuvastatin fig3
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 10 mg Blister Carton (10 x 6 Unit-dose) - rosuvastatin fig4
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg (30 Tablets Bottle) - rosuvastatin fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg Blister Carton (10 x 6 Unit-dose) - rosuvastatin fig6
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 40 mg (30 Tablets Bottle) - rosuvastatin fig7
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 40 mg Blister Carton (10 x 10 Unit-dose) - rosuvastatin fig8
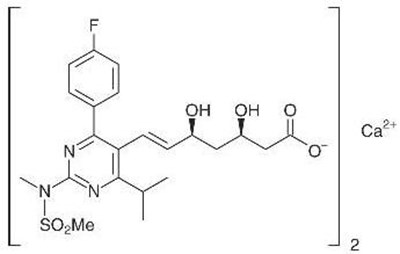
- Chemical Structure - rosuvastatin str
Product Label Images
The following 9 images provide visual information about the product associated with Rosuvastatin Calcium NDC 65862-294 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg Blister Carton (10 x 6 Unit-dose) - rosuvastatin fig2

This is a description of a medication called Rosuvastatin Tablets USP, used for lowering cholesterol levels. The tablets come in a film-coated form and each tablet contains 5mg of rosuvastatin. The medication should be stored away from moisture and out of reach of children, as with all medications. The recommended dosage can be found in the accompanying Prescribing Information. The unit-dose package is child-resistant. The medication is made in India and the NDC code for this specific version is 65862-293-06.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 10 mg (30 Tablets Bottle) - rosuvastatin fig3

Each film-coated tablet contains rosuvastatin calcium USP equivalent to rosuvastatin 10 mg. These tablets are distributed by Aurobindo Pharma USA, Inc with NDC code 65862-294-30. The usual dosage is mentioned in the accompanying prescribing information. The tablets are made in India and a warning is given to keep them out of the reach of children. They should be stored at 20°-25°C and protected from moisture. The text contains a code "TS/DRUGS/19/1993" and some printing instructions in the coding area.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 10 mg Blister Carton (10 x 6 Unit-dose) - rosuvastatin fig4

This is a description of a medication called Rosuvastatin Tablets USP. It is packaged in a unit dose of 50 tablets in 10 blister cards. The tablets are film-coated and contain 10mg of Rosuvastatin calcium USP equivalent to fosuvastatin. The usual dosage guidelines are available in the accompanying prescribing information. The packaging is child-resistant and the medication should be stored between 20° to 25°C (68° to T7°F), with excursion permitted between 15° to 30°C (59° o 86°F), protected from moisture. The manufacturer is Aurobindo Pharma USA, Inc, and the medication was made in India.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg (30 Tablets Bottle) - rosuvastatin fig5

This is a description of the medication Rosuvastatin Tablets USP. Each film-coated tablet contains 20 mg of rosuvastatin calcium USP equivalent to rosuvastatin. It is important to store the medication out of reach of children and at a temperature of 20° to 25°C (68° to 77°F), with excursions permitted to 15° to 30°C (59° to 86°F). The product is distributed by Aurobindo Pharma USA, Inc. and made in India. The accompanying prescribing information should be consulted before use.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg Blister Carton (10 x 6 Unit-dose) - rosuvastatin fig6

This is a description of Rosuvastatin Tablets USP manufactured by Aurobindo Pharma USA Inc. The tablets come in a unit-dose package that is child-resistant with each package containing 6 tablets. Each film-coated tablet has Rosuvastatin calcium USP equivalent to Rosuvastatin 20mg. The normal dosage information is provided in the accompanying prescription information. The tablets should be stored at 20°C to 25°C (68°F to 77°F), with excursions permitted to 15°C to 30°C (59°F to 86°F) and protected from moisture. The package has an NDC number of 65862-295-06, and it is only available with a prescription.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 40 mg (30 Tablets Bottle) - rosuvastatin fig7

Each film-coated tablet contains 10 rosuvastatin 40 mg, which is equivalent to Rosuvastatin calcium USP. It is distributed by Aurobindo Pharma USA, Inc. The usual dosage information can be found in the accompanying Prescribing Information. The tablets are made in India and should be stored at temperatures between 20°C to 25°C (68°F to 77°F) with excursions permitted from 15°C to 30°C (59°F to 86°F). The drug should be kept out of reach of children and protected from moisture. The packaging contains a warning and the printing area must be respected.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 40 mg Blister Carton (10 x 10 Unit-dose) - rosuvastatin fig8

This appears to be the packaging information for Rosuvastatin Tablets sold by Aurobindo Pharma USA. Each film-coated tablet contains Rosuvastatin calcium USP equivalent to Rosuvastatin 40 mg. The package is child-resistant and contains 100 tablets (10 blister cards each containing 10 tablets) with the usual dosage information included in the accompanying Prescribing Information. The product must be stored at temperatures between 20°C and 25°C, protected from moisture. The package was distributed by Aurobindo Pharma USA, Inc. and made in India. The NDC number assigned to this product is 65862-296-10.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.