Product Images Ondansetron
View Photos of Packaging, Labels & Appearance
- ondansetronod-fig1 - ondansetronod fig1
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 4 mg Blister Carton (3 x 10 Unit-dose) - ondansetronod fig2
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 8 mg (6,000 Tablets Bottle) - ondansetronod fig3
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 8 mg Blister Carton (3 x 10 Unit-dose) - ondansetronod fig4
- Chemical Structure - ondansetronod str
Product Label Images
The following 5 images provide visual information about the product associated with Ondansetron NDC 65862-391 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
ondansetronod-fig1 - ondansetronod fig1

This is a description of a medication called Ondansetron Orally Disintegrating Tablets, USP. Each tablet contains Ondansetron USP 4. The package insert gives instructions on usage and administration. The tablets should be stored at a controlled room temperature of 20-25°C. They should not be removed from the package until immediately before use. The medication meets the USP Disintegration Test 2 and should be dispensed in a tight, light-resistant container. The medication is distributed by Aurobindo Pharma USA.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 4 mg Blister Carton (3 x 10 Unit-dose) - ondansetronod fig2

This is a medication description for Ondansetron Orally Disintegrating Tablets, which require a prescription. Each contains 4 grams of Ondansetron USP, and a package should be stored between 20-30°C to maintain shelf life. The package contains 30 tablets, while the dosage and administration instructions should be followed as per the packaging. This particular medication is distributed by Aurobindo Pharma USA, Inc. The text includes a caution that the blister cards should be provided in a child-resistant container, and should not be used if blister packages are open, broken, or have missing medication.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 8 mg (6,000 Tablets Bottle) - ondansetronod fig3

This is the packaging information for Ondansetron Orally Disintegrating Tablets, USP, manufactured by Aurobindo Pharma USA. Each package contains 30 tablets, each containing 8mg of Ondansetron USP. It is recommended to verify the standard dosage and administration from the packaging before use, which is stored at a temperature of 20-25°C (68-77°F). These tablets meet the USP Disintegration Test 2 and should be dispensed from a tight, light-resistant container per the USP guidelines. The package should not be opened until immediately before use. There is no additional information available.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 8 mg Blister Carton (3 x 10 Unit-dose) - ondansetronod fig4
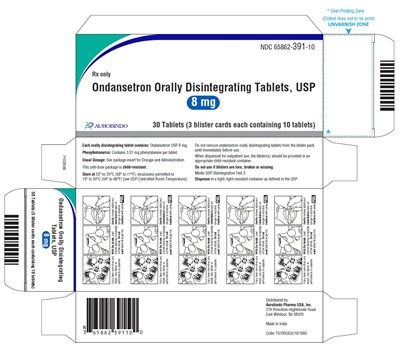
Ondansetron is a medication in the form of orally disintegrating tablets used to prevent nausea and vomiting caused by chemotherapy, radiation therapy, and surgery. This specific package contains 30 tablets and cannot be removed from the blister card until immediately prior to use. Ondansetron tablets should only be used as directed and should not be taken by individuals with phenylketonuria due to the presence of phenylalanine. The medication meets USP Disintegration Test 2 and should be stored at a controlled room temperature between 20-25°C (68-77°F) in a light-resistant container. This medication is manufactured and distributed by Aurobindo Pharma USA.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.