Product Images Divalproex Sodium
View Photos of Packaging, Labels & Appearance
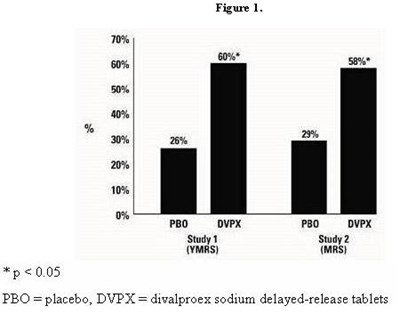
- Figure 1 - divalproex fig1
- divalproex-fig10 - divalproex fig10
- Figure 2 - divalproex fig2
- figure 3 - divalproex fig3
- Figure 4 - divalproex fig4
- divalproex-fig5 - divalproex fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 125 mg Blister Carton (10 x 10 Unit-Dose) - divalproex fig6
- divalproex-fig7 - divalproex fig7
- divalproex-fig8 - divalproex fig8
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 500 mg (100 Tablets Bottle) - divalproex fig9
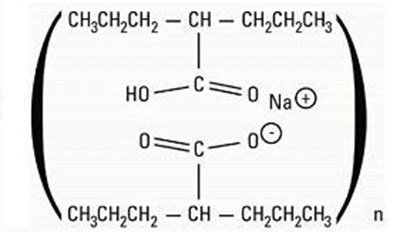
- Chemical Structure - divalproex str
Product Label Images
The following 11 images provide visual information about the product associated with Divalproex Sodium NDC 65862-403 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
divalproex-fig10 - divalproex fig10

This is a medication with the NDC code 65862-403-10 that contains Divalproex Sodium in delayed-release tablets of 500 mg. The usual dosage and packaging information is provided on the package enclosure. The medication guide should be provided separately to each patient by the pharmacist. The package is child-resistant and should be stored at room temperature. The manufacturer is Aurobindo.*
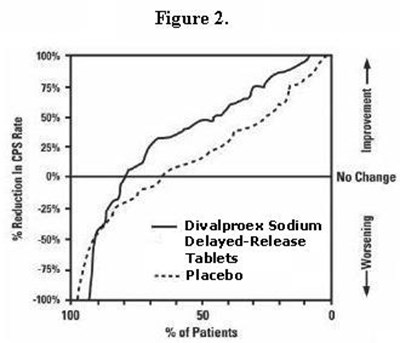
figure 3 - divalproex fig3

This is a chart or a figure showing the % reduction in CPS rate with High Dose and Low Dose on the X-axis and % of Patients on the Y-axis. There are three categories of patients - Improvement, No Change, and Worsening.*
divalproex-fig5 - divalproex fig5

This is a medication label for Divalproex Sodium Delayed-Release Tablets USP. The tablets come in a bottle of 100 and contain 125 mg of valproic acid. The label includes information on dosage, storage temperature, and packaging requirements. The medication is distributed by Aurobindo Pharma USA, Inc. and made in India. NDC number is provided for reference.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 125 mg Blister Carton (10 x 10 Unit-Dose) - divalproex fig6

This is a prescription drug called Divalproex Sodium in the form of delayed-release tablets. The package contains 400 tablets (10 x 10 Unit-Dose) and the standard dosage can be found in the package enclosure. It should be stored between 20° to 25°C with excursions permitted between 15° to 30°C. The medication guide should be provided separately to each patient. It is distributed by Aurobindo Pharma USA, Inc. and it's made in India.*
divalproex-fig8 - divalproex fig8

This is a description of Divalproex Sodium, a medication that comes in the form of delayed-release tablets. Each tablet contains Diviproex sodium USP which is equivalent to Vaproi acid 250mg. The usual dosage should be checked from the package enclosure for prescribing information. It is advised to store at a temperature between 20°C to 25°C (68°F to 77°F) but excursions are permitted from 10°C to 15°C (89°F to 86°F). The unit-dose package is resistible to air and light. The text contains information about the manufacturer of the medicine which is Aurobindo, India. The package contains 100 tablets of 10 x 10 unit-dose each, which is distributable by a pharmacist alongside a medication guide packaged separately for each patient.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 500 mg (100 Tablets Bottle) - divalproex fig9

Each tablet contains 500mg of Divalproex sodium USP, equivalent to valproic acid. It is distributed by Aurobindo Pharma USA, Inc. and comes in an enteric-coated delayed-release form. The tablets should be stored at 20° to 25°C, with excursions up to 15° to 30°C permitted. The medication guide must be provided to each patient, and the bottle should not be accepted if the seal over the bottle opening is broken or missing. This medication is intended for usual dosage, and complete dosing information can be found in the enclosure.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.