Product Images Galantamine
View Photos of Packaging, Labels & Appearance
- Figure 1: Time-Course of the Change From Baseline in ADAS-cog Score for Patients Completing 21 Weeks (5 Months) of Treatment - galantamine fig1
- Figure 10: Time-Course of the Change From Baseline in ADAS-cog Score for Patients Completing 13 Weeks of Treatment - galantamine fig10
- Figure 11: Cumulative Percentage of Patients Completing 13 Weeks of Double-Blind Treatment With Specified Changes from Baseline in ADAS-cog Scores. The Percentages of Randomized Patients Who Completed the Study Were: Placebo 90%, 24 to 32 mg/day 67%. - galantamine fig11
- FIGURE 12 - galantamine fig12
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 4 mg (60 Tablets Bottle) - galantamine fig13
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 8 mg (60 Tablets Bottle) - galantamine fig14
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL -12 mg (60 Tablets Bottle) - galantamine fig15
- Figure 2: Cumulative Percentage of Patients Completing 21 Weeks of Double-Blind Treatment With Specified Changes From Baseline in ADAS-cog Scores. The Percentages of Randomized Patients Who Completed the Study Were: Placebo 84%, 8 mg/day 77%, 16 mg/day 78% and 24 mg/day 78%. - galantamine fig2
- Figure 3: Distribution of CIBIC-plus Ratings at Week 21 - galantamine fig3
- Figure 4: Time-Course of the Change From Baseline in ADAS-cog Score for Patients Completing 26 Weeks of Treatment - galantamine fig4
- FIGURE 5 - galantamine fig5
- Figure 6: Distribution of CIBIC-plus Ratings at Week 26 - galantamine fig6
- Figure 7: Time-Course of the Change From Baseline in ADAS-cog Score for Patients Completing 26 Weeks of Treatment - galantamine fig7
- Figure 8: Cumulative Percentage of Patients Completing 26 Weeks of Double-Blind Treatment With Specified Changes From Baseline in ADAS-cog Scores. The Percentages of Randomized Patients Who Completed the Study Were: Placebo 87%, 24 mg/day 80%, and 32 mg/day 75%. - galantamine fig8
- Figure 9: Distribution of CIBIC-plus Rating at Week 26 - galantamine fig9
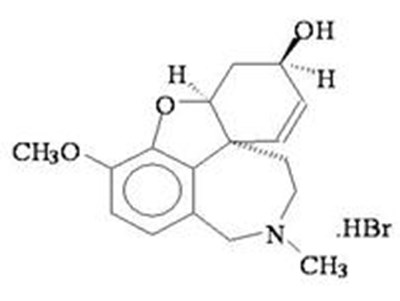
- Chemical Structure - galantamine str
Product Label Images
The following 16 images provide visual information about the product associated with Galantamine NDC 65862-459 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1: Time-Course of the Change From Baseline in ADAS-cog Score for Patients Completing 21 Weeks (5 Months) of Treatment - galantamine fig1

Figure 10: Time-Course of the Change From Baseline in ADAS-cog Score for Patients Completing 13 Weeks of Treatment - galantamine fig10

Figure 11: Cumulative Percentage of Patients Completing 13 Weeks of Double-Blind Treatment With Specified Changes from Baseline in ADAS-cog Scores. The Percentages of Randomized Patients Who Completed the Study Were: Placebo 90%, 24 to 32 mg/day 67%. - galantamine fig11

PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 4 mg (60 Tablets Bottle) - galantamine fig13

Each tablet of Gafantamine hydrobromide USP contains 4 mg of galantamine base. This product is distributed by Aurobindo Pharma USA, Inc. and is made in India. The usual dosage is provided in the accompanying Galantamine product feature. It is recommended to store the tablets at 20-25°C (68-77°F) with excursions permitted to 15-30°C (59-86°F) as per USP Controlled Room Temperature. The package contains 60 tablets and should be kept out of reach of children. No text was available in the overprinting zone coding area.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 8 mg (60 Tablets Bottle) - galantamine fig14

Galantamine is a prescription medication in tablet form, distributed by Aurobindo Pharma USA, Inc. Each tablet contains Galantamine hydrobromide USP equivalent to 8 mg galantamine base. The usual dosage is specified in accompanying product literature. The tablets should be stored at room temperature and protected from excursions in temperature. This medication should be kept out of reach of children. NDC code for the medication is 65862-459-60. The medication is made in India.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL -12 mg (60 Tablets Bottle) - galantamine fig15

This is a description for the medication "Galantami". It is a prescription medication with 60 tablets, each containing 'Galantamine hydrobromide USP, equivalent to 12 mg galantamine base'. The usual dosage information can be found in the product literature. The tablets should be stored at 20° to 25°C (68° to 77°F) with permitted excursions to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. It is important to keep it out of reach of children. This medication is distributed by Aurobindo Pharma USA, Inc., and is made in India.*
Figure 2: Cumulative Percentage of Patients Completing 21 Weeks of Double-Blind Treatment With Specified Changes From Baseline in ADAS-cog Scores. The Percentages of Randomized Patients Who Completed the Study Were: Placebo 84%, 8 mg/day 77%, 16 mg/day 78% and 24 mg/day 78%. - galantamine fig2

Figure 4: Time-Course of the Change From Baseline in ADAS-cog Score for Patients Completing 26 Weeks of Treatment - galantamine fig4

FIGURE 5 - galantamine fig5

The text is showing a chart measuring the cumulative percentage of patients based on their points change related to ADAS-cog from baseline, as well as the dosage of medication administered (placebo, 24mg/day, and 32mg/day). It is not possible to provide further information without context.*
Figure 6: Distribution of CIBIC-plus Ratings at Week 26 - galantamine fig6

This appears to be a graph of the percentage of patients, divided by age group, and their corresponding CIBIC-plus rating (which presumably measures cognitive function). The graph includes three different dosage levels (0, 24mg/day, and 40&24mg/day) and a placebo group. While it is unclear what condition or medication is being examined, it is likely that the graph is showing how these different groups respond to the treatment in terms of improvement or worsening cognitive function.*
Figure 7: Time-Course of the Change From Baseline in ADAS-cog Score for Patients Completing 26 Weeks of Treatment - galantamine fig7

Figure 8: Cumulative Percentage of Patients Completing 26 Weeks of Double-Blind Treatment With Specified Changes From Baseline in ADAS-cog Scores. The Percentages of Randomized Patients Who Completed the Study Were: Placebo 87%, 24 mg/day 80%, and 32 mg/day 75%. - galantamine fig8

Figure 9: Distribution of CIBIC-plus Rating at Week 26 - galantamine fig9

The given text is a table with percentages and the CIBIC-plus Rating. The values in the table indicate the percentage of patients who showed improvement or worsening of their condition under specific doses of medication (specified in mg/day) compared to a placebo. The CIBIC-plus Rating is used for evaluating the effectiveness of the medication on the patient's cognitive and behavioral symptoms. Therefore, the text can be described as a clinical trial report about the effectiveness of a medication on cognitive and behavioral symptoms in patients.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.