Product Images Amoxicillin And Clavulanate Potassium
View Photos of Packaging, Labels & Appearance
Product Label Images
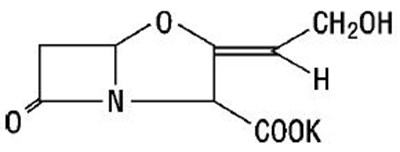
The following 4 images provide visual information about the product associated with Amoxicillin And Clavulanate Potassium NDC 65862-533 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg/28.5 mg* per 5 mL (50 mL Bottle Label) - amoxiclav fig1

This is a prescription drug with the NDC number 65862-533-50. It is Amoxicillin and Clavulanate Potassium, which should be taken orally in suspension form. Once reconstituted, the suspension contains 200 mg of Amoxicillin USP as the trihydrate and 28.5 mg of Clavulanic acid as the clavulanate potassium. It is available in a 50 mL bottle. The directions for mixing are given on the label. The usual dosage is every 12 hours, and it must be refrigerated after opening. The drug should be discarded after 10 days, and the inner seal must be intact for use. It is distributed by Aurobindo Pharma USA, Inc, and made in India.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL – 400 mg/57* mg per 5 mL (50 mL Bottle) - amoxiclav fig2

This is a prescription drug with the NDC code 65862-534-50. It is a powder that is mixed with water to create an oral suspension of Amoxicillin and Clavulanate Potassium, with a concentration of 400 mg/57 mg per 5 mL. It is produced by Aurobindo Pharma USA, Inc. and has a net content of 50 mL when reconstituted. The usual dosage is every 12 hours, and the prescribing information should be consulted. The label warns that it contains phenylalanine and must be refrigerated, and should be discarded after 10 days.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.