Product Images Valsartan And Hydrochlorothiazide
View Photos of Packaging, Labels & Appearance
- Figure 1: Probability of Achieving Systolic Blood Pressure <140 mmHg at Week 8 - valhyctz fig1
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 160 mg/25 mg Blister Carton (10 x 10 Unit-dose) - valhyctz fig10
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 320 mg/12.5 mg (90 Tablets Bottle) - valhyctz fig11
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 320 mg/25 mg (90 Tablets Bottle) - valhyctz fig12
- Figure 2: Probability of Achieving Diastolic Blood Pressure <90 mmHg at Week 8 - valhyctz fig2
- Figure 3: Probability of Achieving Systolic Blood Pressure <130 mmHg at Week 8 - valhyctz fig3
- Figure 4: Probability of Achieving Diastolic Blood Pressure <80 mmHg at Week 8 - valhyctz fig4
- valhyctz-fig5 - valhyctz fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 80 mg/12.5 mg Blister Carton (10 x 10 Unit-dose) - valhyctz fig6
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 160 mg/12.5 mg (90 Tablets Bottle) - valhyctz fig7
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 160 mg/12.5 mg Blister Carton (10 x 10 Unit-dose) - valhyctz fig8
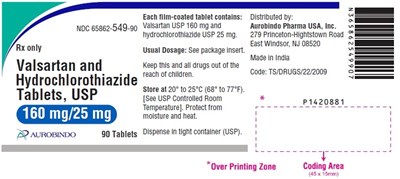
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 160 mg/25 mg (90 Tablets Bottle) - valhyctz fig9
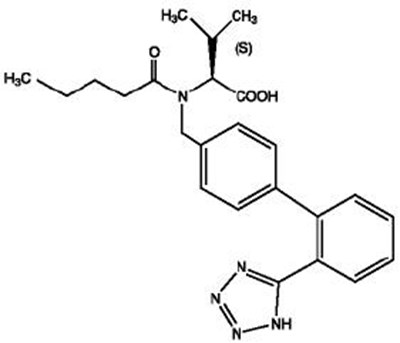
- Chemical Structure1 - valhyctz str1
- Chemical Structure 2 - valhyctz str2
Product Label Images
The following 14 images provide visual information about the product associated with Valsartan And Hydrochlorothiazide NDC 65862-548 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1: Probability of Achieving Systolic Blood Pressure <140 mmHg at Week 8 - valhyctz fig1

This text describes a graph showing the probability of achieving systolic blood pressure below 140mmHg after taking Valsartan and Hydrochlorothiazide medication for 8 weeks. The medication contains 320mg of Valsartan and 25mg of Hydrochlorothiazide. The graph shows the baseline systolic blood pressure at different levels between 145-180 mmHg. The text also includes numerical values used in the graph, 96 and 140, and a code number 2883223288.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 160 mg/25 mg Blister Carton (10 x 10 Unit-dose) - valhyctz fig10

This is a description of a prescription medicine called Valsartan and Hydrochlorothiazide tablets, USP in 160 mg/25 mg strength. The tablets are available in a pack containing 100 units in blister packaging. The tablets contain Valsartan USP in 160mg strength and hydrochlorothiazide USP in 25 mg strength. The tablets are manufactured by Aurobindo Pharma Limited and are for prescription use only. Other than that, the given text does not provide much information and seems to be a combination of codes, instructions, and legal notices.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 320 mg/12.5 mg (90 Tablets Bottle) - valhyctz fig11

This is a medication description for a film-coated tablet containing Valsartan USP 320mg and hydrochlorothiazide USP 12.5mg manufactured by Aurobindo Pharma USA, Inc. with the usual dosage specified in the package insert. It is an Rx-only drug and should be kept out of children's reach and stored at 20-25°C. It comes in a tight container and was distributed in the United States.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 320 mg/25 mg (90 Tablets Bottle) - valhyctz fig12

This is a description of a medication called Valsartan that contains 320mg of USP and hydrochlorothiazide USP 25mg. The medication is manufactured in India by Aurobindo Pharma USA, Inc. Usual dosage is indicated in the package insert and it is available as 90 film-coated tablets per tight container (USP). The text includes a code: TS/IDRUGS/22/2009 and a distributed address in the US: 279 Princeton-Hightstown Road, East Windsor, NJ 08520. However, due to some illegible characters, the output includes some non-English characters, making it difficult to provide an accurate description.*
Figure 2: Probability of Achieving Diastolic Blood Pressure <90 mmHg at Week 8 - valhyctz fig2

This is a figure showing the probability of achieving a diastolic blood pressure of less than 90 mmHg at week 8. There are numerical values ranging from 95 to 105 mmHg along with a baseline value.*
Figure 3: Probability of Achieving Systolic Blood Pressure <130 mmHg at Week 8 - valhyctz fig3

The text describes a figure labeled Figure 3 that shows the probability of achieving a systolic blood pressure (SBP) of less than 130 mmHg at week 8 for patients treated with Valsartan and Hydrochlorothiazide at a certain dosage, as well as a placebo group. The baseline SBP (mmHg) ranges from 140 to 180.*
Figure 4: Probability of Achieving Diastolic Blood Pressure <80 mmHg at Week 8 - valhyctz fig4

This appears to be a description of a graph showing the probability of achieving a diastolic blood pressure less than 80 mmHg after taking Valsartan and Hydrochlorothiazide at certain dosages compared to a placebo. The dosages listed are Valsartan 320 mg and Hydrochlorothiazide 25 mg. The graph shows the probability at week 8 of achieving a blood pressure less than 80 mmHg.*
valhyctz-fig5 - valhyctz fig5

This is a description of a drug called Valsartan Hydrochlorothiazide. It comes in the form of 90 tablets, with each film-coated tablet containing Valsartan USP 80 mg and hydrochlorothiazide USP 12.5 mg. This drug is distributed by Aurobindo Pharma USA, Inc. and is available only by prescription (Rx only). The drug should be kept at 20° to 25°C to ensure preservation, and should be stored in a tight container to prevent spoilage. This drug is not for use by children and the usual dosage should be consulted from the package insert.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 80 mg/12.5 mg Blister Carton (10 x 10 Unit-dose) - valhyctz fig6

This is a detailed description of medication tablets named Valsartan and Hydrochlorothiazide Tablets, USP. The pack contains 100 unit dose tablets, and each tablet is film-coated containing 80 mg of Valsartan USP and 12.5 mg of Hydrochlorothiazide USP. It is mentioned that the dotted lines should not be printed, and there is also a warning that Rx use only, which implies that these tablets should be used only with the instructions of a certified medical professional. However, there are some unreadable or unidentifiable characters in the text.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 160 mg/12.5 mg (90 Tablets Bottle) - valhyctz fig7

This is a description of a medication bottle for Valsartan and Hydrochlorothiazide tablets in 160mg/12.5mg strength, manufactured by Aurobindo Pharma USA. The packaging contains 90 tablets. Recommended dosage and storage information is available on the package insert. The medication should be kept out of reach of children, stored at 68-77 degrees Fahrenheit and protected from heat and moisture. The container has a coding area for printing.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 160 mg/12.5 mg Blister Carton (10 x 10 Unit-dose) - valhyctz fig8

This description is not available as the text includes many nonsensical words and fragmented sentences.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.