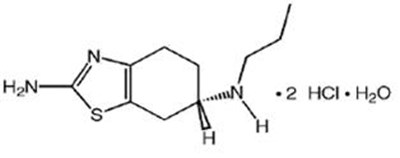
Product Images Pramipexole Dihydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Pramipexole Dihydrochloride NDC 65862-605 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 0.125 mg (90 Tablet Bottle) - pramipexole fig1

Each tablet of this medication contains 0125mg of pramipexole dihydrochloride monohydrate, which is equivalent to 0.118mg of pramipexole dihydrochloride USP. The medication is manufactured in India by Hurobindo Pharma USA, Inc. and is supplied in a tight, light-resistant container. The recommended dosage and prescribing information are provided with the medication. It should be stored at 20°C to 25°C and protected from light. This medication should be kept out of the reach of children. The remaining text is not-readable.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 0.5 mg (90 Tablet Bottle) - pramipexole fig3

This is a description for Pramipexole Dihydrochloride Tablets, a prescription medication containing 0.5 mg of pramipexole dihydrochloride monohydrate in each tablet, distributed by Aurobindo Pharma USA, Inc. It is used for treatment purposes, and the usual dosage can be read in the accompanying prescribing information. It should be stored at 20° to 25°C and protected from light. The prescription should be dispensed in a tight, light-resistant container and kept out of reach of children. NDC 65862-606-90 is the National Drug Code for this medication.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 0.75 mg (90 Tablet Bottle) - pramipexole fig4

Each tablet contains 0.75 mg of pramipexole dihydrochloride monohydrate, equivalent to 0.705 mg of pramipexole. It is distributed by Aurobindo Pharma USA, Inc with NDC 65862-607-90. The tablets should be stored at 20° to 25°C (68° to 77°F) and protected from light. The drug's usual dosage should be read on the accompanying prescribing information. The tablets should be kept out of reach of children. There appears to be some overlapping and missing text.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 1 mg (90 Tablet Bottle) - pramipexole fig5

This is a product description for Pramipexole Dihydrochloride Tablets with NDC 65862-608-90. Each tablet contains 1 mg pramipexole dinydrochloride monohydrate equivalent to 0.94 mg pramipexole dinydrochioride USP. It is distributed by Aurobindo Pharma USA Inc. and it is recommended to dispense in a tight, light-resistant container. The product is made in India and the usual dosage instructions should be read before use. It should be stored at 20°-25°C and protected from light, and kept out of reach of children. The remaining text is not available for evaluation.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 1.5 mg (90 Tablets Bottle) - pramipexole fig6

Each tablet contains 1.5mg of pramipexole dihydrochloride monohydrate, equivalent to 1.41mg pramipexole dihydrochloride USP. It is distributed by Aurohindo Pharma USA, Inc. and bears NDC 65862-609-90. It is only available through prescription and should be dispensed in a tight, light-resistant container. The usual dosage should be read through the accompanying prescribing information. The tablets should be stored in a controlled room temperature of 20° to 25°C while being protected from light. It should be kept away from the reach of children. The printing zone and coded area are indicated and must be observed. The product is made in India.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.