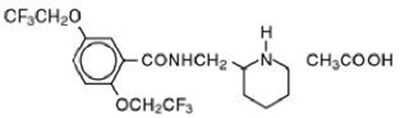
Product Images Flecainide Acetate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Flecainide Acetate NDC 65862-623 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg (30 Tablets Bottle) - flecainide fig1

This is a bulk package of Flecainide Acetate tablets, USP that contains 30 tablets with each tablet having 50 mg of Flecainide acetate USP. It is for Rx only and the usual dosage can be found in the package insert. The tablets should be dispensed in a tight, light-resistant container and stored at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Distributed by Aurobindo Pharma USA, nc. in East Windsor, NJ 08520.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg (30 Tablets Bottle) - flecainide fig2

Each tablet contains Flecainide Acetate USP 100 mg, and is distributed by Aurobindo Pharma USA, Inc. The tablets should be stored in a tight, light-resistant container at room temperature. The package insert should be consulted for dosage instructions. The product is dispensed in a package containing 30 tablets. Made in India.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 150 mg (30 Tablets Bottle) - flecainide fig3

Each tablet contains 150 mg of Flecainide acetate USP. It is distributed by Aurobindo Pharma USA, Inc. The product is dispensed in a tight, light-resistant container and should be stored at 20°C to 25°C. Usual dosage information can be found in the package insert. The product comes in a package of 30 tablets with the NDC code of 65862-623-30. The code for this product is TS/DRUGS/22/2009.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.