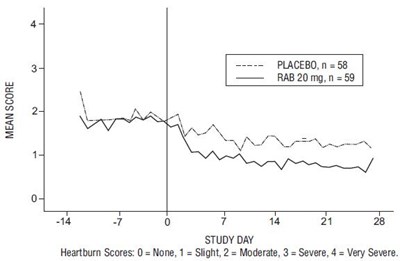
Rabeprazole Fig1
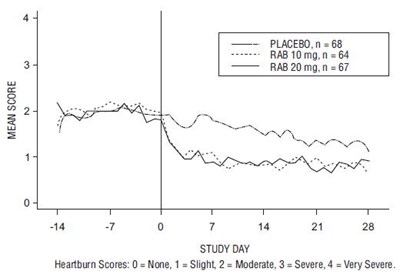
The text provides mean scores for different treatments on heartburn among a sample of 68 people on placebo, 64 people on RAB 10mg, and 67 people on RAB 20mg. The heartburn scores are rated on a scale of 0 to 4, with 0 indicating no heartburn, and 4 indicating very severe heartburn. This information can be useful for evaluating the effectiveness of these treatments in reducing heartburn symptoms.*