Product Images Cefixime
View Photos of Packaging, Labels & Appearance
Product Label Images
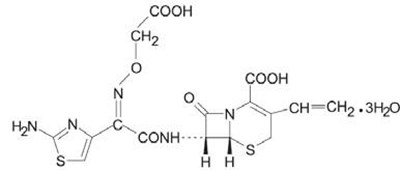
The following 3 images provide visual information about the product associated with Cefixime NDC 65862-751 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg/5 mL (50 mL Bottle) - cefixime fig1

Cefixime for Oral Suspension is a prescription medicine in the form of a reconstituted suspension for oral use only, produced by Aurbindo Pharma USA, Inc. The drug powder must be stored at 20° to 25°C (68° to 77°F) prior to reconstitution, and the reconstituted suspension should be stored under refrigeration or at room temperature. One bottle of the suspension is intended for dispensing and should only be used if the seal is intact. The usual dosage is indicated in the package insert and each mL (measured with a spoon) of the reconstituted suspension contains cefixime trihydrate equivalent to 100 mg of cefixime. The net contents of the bottle contains 1 gram of cefixime trihydrate. After 14 days, discard any unused portion of the suspension. The code "TS/DRUGSTE86" and a date of constitution may be printed in the overprinting zone, while a dotted line in the coding area should not be printed.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.