Product Images Pregabalin
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 30 images provide visual information about the product associated with Pregabalin NDC 65862-760 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
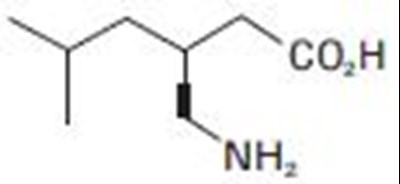
Pregabalin equation - pregabalin equ
This text appears to be formulas used in the medical field to calculate certain values. The first formula calculates something based on the value of someone's age (in years) multiplied by their weight (in kg), where the result is not specified. The second formula calculates a value for CLCr (possibly creatinine clearance) based on the value of someone's serum creatinine level (in mg/dL) with a multiplication factor of 0.85 for female patients.*
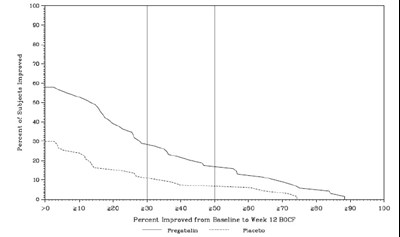
Figure 1: Patients Achieving Various Levels of Improvement in Pain Intensity – Study DPN 1 - pregabalin fig1

The text describes a figure called "Patients Achieving Various Levels of Improvement in Pain Intensity" from a study named "DPN 1". The figure shows the percentage of patients who achieved different levels of pain improvement. The figure includes four groups: Pregabalin 200 mg three times a day, Pregabalin 100 mg three times a day, Pregabalin 25 mg three times a day, and placebo. The percentage of patients improved ranges from 0 to 100.*
Figure 10 - pregabalin fig10

The figure shows the Kaplan-Meier analysis of the time to loss of therapeutic response for a fibromyalgia study F2. The percentage of subjects without LTR is estimated over time for the pregabalin and placebo groups. Not available.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 25 mg (90 Capsules Bottle) - pregabalin fig13

This is a description of Pregabalin capsules with a medication code of DG 6585275890. The medication is distributed by Aurobindo Pharma USA and each capsule contains 25 mg of Pregabain USP. The dosage information can be found in the accompanying prescribing information and the medication should be stored at a temperature range of 20° to 25° Celsius. It should be dispensed in a tight container. The remaining text is not readable.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 25 mg 100 (10 x 10) Unit-dose Capsules - pregabalin fig14

Each capsule contains 25mg of Pregabalin USP. The prescribing information should be consulted for the usual dosage. The capsules should be stored in tight containers at a temperature range of 20°C to 25°C. The unit-dose package is not child-resistant; dispensing for outpatient use requires a child-resistant container. The medication guide should be provided to each patient. The product is distributed by Aurobindo Pharma USA Inc. under Code TS/DRUGS/22/2009 and produced in India. No further information is available.*
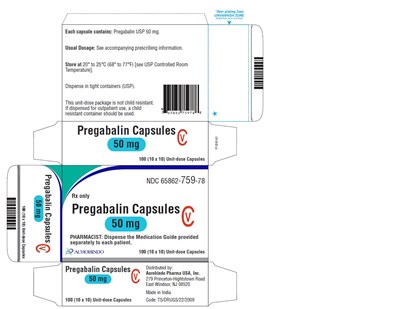
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg (90 Capsules Bottle) - pregabalin fig15

This is the description of a medicine - Pregabalin USP with a strength of 50mg in each capsule. It is distributed by Aurobindo Pharma USA, Inc. The medication needs to be dispensed by the pharmacist in tight containers as per USP guidelines. The dosage of the medicine is mentioned in the accompanying Pregabalin prescribing information. The medicine is available in the form of capsules and should be stored at a temperature between 20° to 25°C. The provided information also includes the name, address and NDC code of the manufacturer.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 75 mg 100 (10 x 10) Unit-dose Capsules - pregabalin fig18

This is a description of Pregabalin Capsules medication in a unit-dose package of 100 capsules. Each capsule contains 75mg of Pregabalin USP. It is advised to store the medication at a controlled room temperature of 20°C to 25°C (68°F to 77°F) and dispense in tight containers. The prescription dosage should be referred to for administration instructions. If dispensed for outpatient use, a child-resistant container should be used. The capsules were distributed by a company called Aurobindo and made in India. There is additional text that is not readable due to errors.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg (90 Capsules Bottle) - pregabalin fig19

This is a description of a medication called Pregabalin, which comes in capsules containing 100mg of Pregabalin USP. The medication is only available with a prescription (Rx only), and each patient should receive a medication guide. The usual dosage is indicated in the accompanying prescribing information. The medication should be stored in a controlled room temperature of 20° to 25°C (68° to 77°F), and dispensed in tight containers (USP). It is distributed by Aurobindo Pharma USA, Inc. from East Windsor, NJ and made in India.*
Figure 2: Patients Achieving Various Levels of Improvement in Pain Intensity – Study - pregabalin fig2

The figure shows the percentage of patients who achieved various levels of improvement in pain intensity in Study DPN 2, comparing Pregabalin 100 mg three times a day to a Placebo group. The x-axis represents the percentage of improvement in pain from baseline, and the y-axis represents the percentage of patients improved.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg 100 (10 x 10) Unit-dose Capsules - pregabalin fig20

This is a description of Pregabalin Capsules containing 100 mg of the active ingredient. The usual dosage information can be found in the accompanying prescribing information. The capsules should be stored in tight containers at a temperature of 20-25°C. The package is not child resistant, so a child-resistant container should be used if dispensed for outpatient use. The information includes the unit-dose package details along with its barcode and drug code. The product is manufactured in India and distributed in the US by Pramaush Inc.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 150 mg (90 Capsules Bottle) - pregabalin fig21

This is a description of a medication called Pregabalin, which comes in capsule form. The medication is only available through prescription and comes with a Medication Guide for each patient. The capsules are made by the pharmaceutical company Aurobindo Pharma USA, Inc. Each capsule contains 150mg of Pregabalin USP. The recommended dosage can be found in the accompanying prescribing information. The medication should be stored in a controlled room temperature environment and dispensed in a tight container. The label also includes a code and address for the manufacturer. The text also includes a note about not printing the dotted lines in a specific area.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 150 mg 100 (10 x 10) Unit-dose Capsules - pregabalin fig22

This is a description of Pregabalin Capsules containing 150mg of the active ingredient Pregabalin USP. The text provides information on the recommended dosage and storage conditions. It also notes that if the capsules are dispensed for outpatient use, a child-resistant container should be used. The package includes 100 unit-dose capsules and the medication guide should be provided separately to each patient. The distributor is R0 ush e m V in New Jersey, and the capsules are made in India.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg (90 Capsules Bottle) - pregabalin fig23

Each capsule of pregabalin USP contains 200 mg and is distributed by Aurobindo Pharma USA, Inc. The usual dosage information can be found in the accompanying prescribing information. Pharmacists are advised to dispense the Medication Guide separately to each patient, and the medication should be stored at 20° to 25°C (68° to 77°F). It must be dispensed in tight containers and the dotted lines in the overprinting zone should not be printed.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg 100 (10 x 10) Unit-dose Capsules - pregabalin fig24

This is a label for Pregabalin Capsules containing 200mg of Pregabalin USP. The usual dosage has to be seen in the attached prescribing information. It needs to be stored at 20° to 25°C. It should be dispensed in tight containers and should be kept child-resistant. The package contains 100 unit-dose capsules that are distributed by Aurobindo Pharma USA, Inc. The label has the NDC code 65862-763-78 and it is meant for outpatient use only. The medication also comes with a Medication Guide for patients.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 225 mg 100 (10 x 10) Unit-dose Capsules - pregabalin fig26

This is a description of Pregabalin capsules containing 225mg of the medication. The capsules are recommended for use as per accompanying prescribing information. The medication should be stored at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature] and dispensed in a tight container (USP). The capsules are sold in a unit-dose package of 100 (10 x 10) capsules with an NDC code of 65862-764-78. The medication is distributed by Aurobindo Pharma USA, Inc. and made in India. There is additional text that appears to be unrelated and potentially randomly generated.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 300 mg (90 Capsules Bottle) - pregabalin fig27

The text is a prescription drug label for Pregabalin capsules manufactured by Aurobindo Pharma USA, Inc. It includes information for pharmacists to dispense the medication, such as the usual dosage, storage conditions and precautions. Also, it contains the NDC code, number of capsules, strength, and a unique batch code. The label advises the pharmacist to dispense the medication guide to each patient.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 300 mg 100 (10 x 10) Unit-dose Capsules - pregabalin fig28

This is a description of Pregabalin capsules, including their dosage, storage requirements, and packaging. The capsules contain 300mg of Pregabalin USP and are intended for outpatient use. They should be stored in a container at 20-25°C and dispensed in a tight container. The packaging is not child-resistant, and if used for outpatient purposes, a child-resistant container should be used. Additionally, a prescription is required to obtain these capsules, and the accompanying medication guide should be provided to each patient. The capsules are manufactured by Aurobindo Pharma USA and are made in India.*
Figure 3: Patients Achieving Various Levels of Improvement in Pain Intensity – Study PHN 1 - pregabalin fig3

This is a figure showing the percentage of patients who achieved various levels of improvement in pain intensity in study PHN 1. The data includes patients taking pregabalin at different dosages (75 mg, 150 mg, and 300 mg, two times a day) as well as those receiving placebo. The percentage of patients who experienced improvement in pain intensity from baseline ranges from 10% to 100%.*
Figure 4: Patients Achieving Various Levels of Improvement in Pain Intensity – Study PHN 2 - pregabalin fig4

This is a graph depicting the percentage of patients achieving various levels of improvement in pain intensity in Study PHN 2. The X-axis shows the percent improvement in pain from baseline, and the Y-axis shows the percentage of patients improved. Three lines are shown on the graph: one for patients taking Pregabalin 200 mg three times a day, one for patients taking Pregabalin 100 mg three times a day, and one for patients taking Placebo.*
Figure 5: Patients Achieving Various Levels of Improvement in Pain Intensity – Study PHN 3 - pregabalin fig5

The text describes a figure showing the percentage of patients achieving different levels of improvement in pain intensity in a study named PHN 3, while being treated with Pregabalin at 50 mg and 100 mg three times a day, or a placebo. The X-axis shows the percentage of patients improved and the Y-axis shows the percent improvement in pain from baseline.*
figure6 - pregabalin fig6

The text seems to be a set of tables with some numbers and symbols, followed by the text "statistically significant vs placebo". Therefore, the description of the content is related to a comparison between different studies or experiments on some subject, possibly related to a medical or scientific research. More context is needed to provide a more accurate description.*
Figure 8 - pregabalin fig8

The text seems to represent a table or chart with several percentages under the label "Responder Rate". However, without additional context or headings, it is not possible to determine the meaning or context of the percentages provided.*
Figure 9 - pregabalin fig9

The figure shows the percentage of patients experiencing different levels of improvement in pain intensity in a study conducted for fibromyalgia. Four groups were tested: patients taking a daily dose of 600 mg of Pregabalin, those taking a daily dose of 450 mg of Pregabalin, those taking a daily dose of 300 mg of Pregabalin, and those taking a placebo. The x-axis reports the percentage of improvement in pain from the baseline, while the y-axis reports the percentage of patients that experienced that level of improvement. The graph shows that the higher the dose of Pregabalin, the more patients experienced an improvement in pain intensity.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.