Product Images Esomeprazole Magnesium
View Photos of Packaging, Labels & Appearance
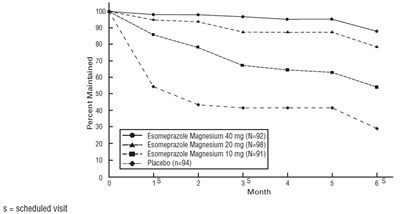
- Figure 3: Maintenance of Healing Rates by Month (Study 178) - esomeprazole fig1
- Figure 2: Maintenance of Healing Rates by Month (Study 177) - esomeprazole fig2
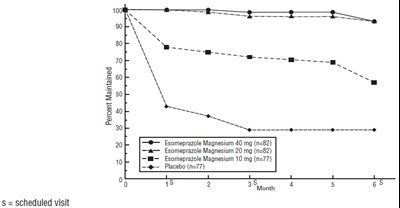
- Figure 4: Percent of Patients Symptom-Free of Heartburn by Day (Study 225) - esomeprazole fig3
- Figure 5: Percent of Patients Symptom-Free of Heartburn by Day (Study 226) - esomeprazole fig4
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg (30 Capsules Bottle) - esomeprazole fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 MG (10 X 10) UNIT-DOSE CAPSULES - esomeprazole fig6
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 40 MG (30 CAPSULES BOTTLE) - esomeprazole fig7
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 40 MG (10 X 10) UNIT-DOSE CAPSULES - esomeprazole fig8
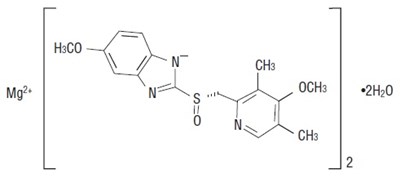
- Chemical Structure - esomeprazole str
Product Label Images
The following 9 images provide visual information about the product associated with Esomeprazole Magnesium NDC 65862-784 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 4: Percent of Patients Symptom-Free of Heartburn by Day (Study 225) - esomeprazole fig3

This is a clinical trial result showing the percentage of patients who were symptom-free after using Esomeprazole Magnesium Delayed-Release Capsules 40 mg or 20 mg compared to those who took a placebo. The trial lasted for 21 days, and the symptom-free rate was 100% for both doses of the medication, while the placebo showed a lower percentage.*
Figure 5: Percent of Patients Symptom-Free of Heartburn by Day (Study 226) - esomeprazole fig4

This appears to be a graph but the data is not sufficient for generating a useful description.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 MG (10 X 10) UNIT-DOSE CAPSULES - esomeprazole fig6

Esomeprazole Magnesium Delayed-Release Capsules USP are a medication that each contain 20 mg of esomeprazole magnesium dihydrate. The capsules are intended for adult usage as directed on the package insert and must be stored at controlled room temperature between 20-25°C (68-77°F). The packaging is non-child resistant and is not safe for dispensing to patients who need child-proof containers. The medication guide is to be provided to each patient by their pharmacist. The capsules are distributed by Aurobindo Pharma in the USA and were manufactured in India.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 40 MG (30 CAPSULES BOTTLE) - esomeprazole fig7

This is a prescription medicine with NDC number 65862-784-30. It contains Esomeprazole Magnesium in the form of Delayed-Release Capsules USP, equivalent to 40 mg of esomeprazole. The brand Aurobindo distributes the 30 capsules, and the medication guide should be provided to each patient. There is additional information regarding dosage recommendations and storage instructions. There is also a printed warning about a "Zone Coding Area" that should not be printed over.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 40 MG (10 X 10) UNIT-DOSE CAPSULES - esomeprazole fig8

This is a medication called Esomeprazole Magnesium which comes in 100 unit-dose capsules. Each capsule contains 40 mg of Esomeprazole magnesium dihydrate equivalent. The medication is for adult use only and the package insert should be consulted for proper dosage information. It should be stored at controlled room temperature between 20° to 25°C (68° to 77°F). The container must be tightly closed and is not child resistant. The medication guide should be provided separately to each patient. The capsules are distributed by Aurobindo Pharma USA, Inc. and made in India.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.