Product Images Amlodipine, Valsartan And Hydrochlorothiazide
View Photos of Packaging, Labels & Appearance
- figure1 - amlovalhctz fig1
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 10 mg/320 mg/25 mg (30 Tablets Bottle) - amlovalhctz fig10
- figure2 - amlovalhctz fig2
- figure3 - amlovalhctz fig3
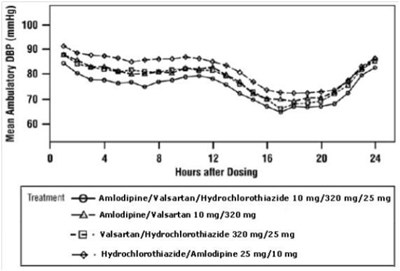
- figure4 - amlovalhctz fig4
- figure5 - amlovalhctz fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg/160 mg/12.5 mg (30 Tablets Bottle) - amlovalhctz fig6
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL -10 mg/160 mg/12.5 mg (30 Tablets Bottle) - amlovalhctz fig7
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg/160 mg/25 mg (30 Tablets Bottle) - amlovalhctz fig8
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 10 mg/160 mg/25 mg (30 Tablets Bottle) - amlovalhctz fig9
- Amlodipine Molecular Structure - amlovalhctz str1
- Valsartan Molecular Structure - amlovalhctz str2
- Hydrochlorothiazide Molecular Structure - amlovalhctz str3
Product Label Images
The following 13 images provide visual information about the product associated with Amlodipine, Valsartan And Hydrochlorothiazide NDC 65862-834 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure1 - amlovalhctz fig1

This appears to be medical information describing dosages and combinations of medications used to treat hypertension.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 10 mg/320 mg/25 mg (30 Tablets Bottle) - amlovalhctz fig10

This appears to be a description of a medication called Amlodipine, Valsartan, and Hydrochlorothiazide which comes in a bottle labeled "Exchfimcostd bl comtalns" with a dosage of "Dt B8z 838.80 ooy 5P oo Prama U". The medication is in tablet form, with a label "Tablets USP Tl I i" and a code of "Cot: TS/DRUGS/2222000" and "P1422175". The label also mentions to keep the medication in a safe place and not to use past the expiration date. The bottle appears to be manufactured by Aurobindo, but there is a pending test for organic impurities.*
figure3 - amlovalhctz fig3

This is a tabular form showing Mean Siting SBP (mmHg) values ranging from 120 to 180. There are six weeks of data displayed on the horizontal axis with corresponding treatments listed underneath. The treatments include Amlodipine/Valsartan/Hydrochlorothiazide 10mg/320mg/25mg, Amlodipine/Valsartan 10mg/320mg, Valsartan/Hydrochlorothiazide 320mg/25mg and Hydrochlorothiazide/Amlodipine 25mg/10mg.*
figure5 - amlovalhctz fig5

This is a graph showing the mean Ambulatory SBP (Systolic Blood Pressure) over time, up to 24 hours after dosing with different treatments. The treatments include a combination of Amlodipine, Valsartan, and Hydrochlorothiazide in different doses. The graph compares three different treatment options. The x-axis shows Hours after Dosing, and the y-axis shows the SBP values. It is useful for comparing the efficacy of different antihypertensive treatments.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg/160 mg/12.5 mg (30 Tablets Bottle) - amlovalhctz fig6

A combination medication containing Amlodipine, Valsartan, and Hydrochlorothiazide is available in the form of tablets. It is manufactured by Aurobindo and each tablet contains 5 mg/160 mg/12.5 mg of the medicines respectively. The product has a unique identifier number. The rest of the text does not appear to be useful information.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg/160 mg/25 mg (30 Tablets Bottle) - amlovalhctz fig8

This appears to be a partial description of pharmaceutical products. It includes the name of a drug "Amlodipine, Valsartan, and Hydrochlorothiazide Tablets USP" and some of its specifications, such as weight and storage conditions. The text also includes a manufacturer's name and address. However, the content is not clear and complete enough to provide a useful description in its current form.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.