Product Images Iclevia
View Photos of Packaging, Labels & Appearance
- Figure A - levo ethi fig1
- Figure B - levo ethi fig2
- Figure 3 - levo ethi fig3
- Figure 4 - levo ethi fig4
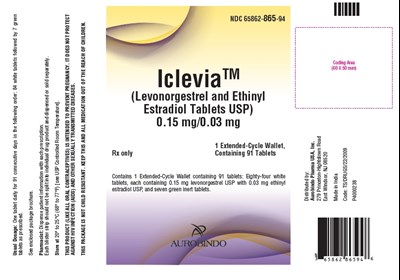
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 0.15 mg/0.03 mg (91 Tablets Carton) - levo ethi fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 0.15 mg/0.03 mg (91 Tablets Pouch) - levo ethi fig6
- levo ethi fig7
- levo ethi fig8
- levo ethi fig9
- structure1 - levo ethi str1
- structure2 - levo ethi str2
Product Label Images
The following 11 images provide visual information about the product associated with Iclevia NDC 65862-865 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 3 - levo ethi fig3

The given text provides information about various studies conducted comparing the use of combination oral contraceptives (COC) with non-use. These studies include NICHD Women's Care Study, French E3N cohort Study, Shanghai Women's Health Study, The Nurses’ Health Study I, and Oxford Family Planning Study. The text also includes statistical measures such as Odds Ratio (OR), Relative Risk (RR), and Hazard Ratio (HR) for some of the studies. Additionally, the text mentions Effect Estimate but it is not clear what it refers to.*
Figure 4 - levo ethi fig4
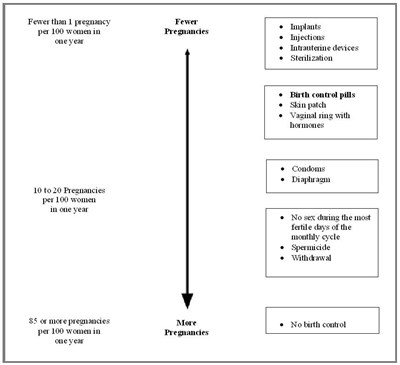
This document provides different methods of contraception for women and the percentage of effectiveness in preventing pregnancy in one year. It shows the range from fewer than 1 pregnancy per 100 women to 85 or more pregnancies per 100 women. The methods include implants, injections, intrauterine devices, sterilization, birth control pills, skin patch, vaginal ring with hormones, condoms, diaphragm, no intercourse during the most fertile days of the monthly cycle, spermicide, withdrawal, and no birth control.*
levo ethi fig8
Not available. The given text seems to be a mixture of random characters, symbols, and words with no clear meaning or context.*
levo ethi fig9

This is a description of a medication called Iclevia™ which is a combination of levonorgestrel and ethinyl estradiol in tablet form. It comes in three extended-cycle wallets, each containing 91 tablets, with 84 white tablets containing 0.15mg levonorgestrel USP and 0.03mg ethinyl estradiol USP and seven green inert tablets. It is used to prevent pregnancy when taken in the prescribed daily doses for 91 consecutive days. It does not protect against sexually transmitted diseases such as HIV. The medication should be stored at room temperature, between 20°C to 25°C. The medication is distributed by Aurobindo Pharma USA, Inc.*
structure1 - levo ethi str1

Levonorgestrel is a chemical compound with a molecular weight of 3124 g/mol.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.





